| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Endothelin-1 receptor |
|---|
| Ligand | BDBM50288013 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_65464 (CHEMBL682104) |
|---|
| IC50 | 2.8±n/a nM |
|---|
| Citation |  Chan, MF; Raju, B; Kois, A; Castillo, RS; Verner, EJ; Wu, C; Hwang, E; Okun, I; Stavros, F; Balaji, VN Halogen substitution at the isoxazole ring enhances the activity of N-(isoxazolyl)sulfonamide endothelin antagonists Bioorg Med Chem Lett6:2393-2398 (1996) Article Chan, MF; Raju, B; Kois, A; Castillo, RS; Verner, EJ; Wu, C; Hwang, E; Okun, I; Stavros, F; Balaji, VN Halogen substitution at the isoxazole ring enhances the activity of N-(isoxazolyl)sulfonamide endothelin antagonists Bioorg Med Chem Lett6:2393-2398 (1996) Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Endothelin-1 receptor |
|---|
| Name: | Endothelin-1 receptor |
|---|
| Synonyms: | EDNRA | EDNRA_HUMAN | ET-A | ETA | ETA-R | ETRA | Endothelin receptor type A | Endothelin receptor, ET-A/ET-B | hET-AR |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 48736.88 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P25101 |
|---|
| Residue: | 427 |
|---|
| Sequence: | METLCLRASFWLALVGCVISDNPERYSTNLSNHVDDFTTFRGTELSFLVTTHQPTNLVLP
SNGSMHNYCPQQTKITSAFKYINTVISCTIFIVGMVGNATLLRIIYQNKCMRNGPNALIA
SLALGDLIYVVIDLPINVFKLLAGRWPFDHNDFGVFLCKLFPFLQKSSVGITVLNLCALS
VDRYRAVASWSRVQGIGIPLVTAIEIVSIWILSFILAIPEAIGFVMVPFEYRGEQHKTCM
LNATSKFMEFYQDVKDWWLFGFYFCMPLVCTAIFYTLMTCEMLNRRNGSLRIALSEHLKQ
RREVAKTVFCLVVIFALCWFPLHLSRILKKTVYNEMDKNRCELLSFLLLMDYIGINLATM
NSCINPIALYFVSKKFKNCFQSCLCCCCYQSKSLMTSVPMNGTSIQWKNHDQNNHNTDRS
SHKDSMN
|
|
|
|---|
| BDBM50288013 |
|---|
| n/a |
|---|
| Name | BDBM50288013 |
|---|
| Synonyms: | 5-Bromo-N-(4-bromo-3-methyl-isoxazol-5-yl)-2-ethyl-benzenesulfonamide | CHEMBL309692 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C12H12Br2N2O3S |
|---|
| Mol. Mass. | 424.108 |
|---|
| SMILES | CCc1ccc(Br)cc1S(=O)(=O)Nc1onc(C)c1Br |
|---|
| Structure | 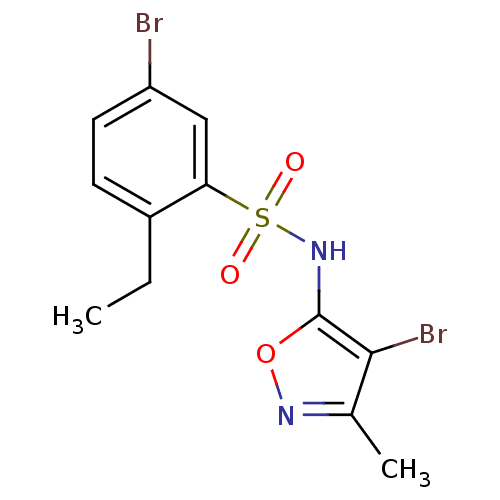
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chan, MF; Raju, B; Kois, A; Castillo, RS; Verner, EJ; Wu, C; Hwang, E; Okun, I; Stavros, F; Balaji, VN Halogen substitution at the isoxazole ring enhances the activity of N-(isoxazolyl)sulfonamide endothelin antagonists Bioorg Med Chem Lett6:2393-2398 (1996) Article
Chan, MF; Raju, B; Kois, A; Castillo, RS; Verner, EJ; Wu, C; Hwang, E; Okun, I; Stavros, F; Balaji, VN Halogen substitution at the isoxazole ring enhances the activity of N-(isoxazolyl)sulfonamide endothelin antagonists Bioorg Med Chem Lett6:2393-2398 (1996) Article