| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Muscarinic acetylcholine receptor M2 |
|---|
| Ligand | BDBM50228363 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_495765 (CHEMBL1007884) |
|---|
| IC50 | 6280±n/a nM |
|---|
| Citation |  Vu, CB; Bemis, JE; Disch, JS; Ng, PY; Nunes, JJ; Milne, JC; Carney, DP; Lynch, AV; Smith, JJ; Lavu, S; Lambert, PD; Gagne, DJ; Jirousek, MR; Schenk, S; Olefsky, JM; Perni, RB Discovery of imidazo[1,2-b]thiazole derivatives as novel SIRT1 activators. J Med Chem52:1275-83 (2010) [PubMed] Article Vu, CB; Bemis, JE; Disch, JS; Ng, PY; Nunes, JJ; Milne, JC; Carney, DP; Lynch, AV; Smith, JJ; Lavu, S; Lambert, PD; Gagne, DJ; Jirousek, MR; Schenk, S; Olefsky, JM; Perni, RB Discovery of imidazo[1,2-b]thiazole derivatives as novel SIRT1 activators. J Med Chem52:1275-83 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Muscarinic acetylcholine receptor M2 |
|---|
| Name: | Muscarinic acetylcholine receptor M2 |
|---|
| Synonyms: | ACM2_HUMAN | CHRM2 | Cholinergic, muscarinic M2 | Muscarinic acetylcholine receptor M2 and M4 | Muscarinic acetylcholine receptor M2 and M5 | RecName: Full=Muscarinic acetylcholine receptor M2 |
|---|
| Type: | GPCR |
|---|
| Mol. Mass.: | 51730.61 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P08172 |
|---|
| Residue: | 466 |
|---|
| Sequence: | MNNSTNSSNNSLALTSPYKTFEVVFIVLVAGSLSLVTIIGNILVMVSIKVNRHLQTVNNY
FLFSLACADLIIGVFSMNLYTLYTVIGYWPLGPVVCDLWLALDYVVSNASVMNLLIISFD
RYFCVTKPLTYPVKRTTKMAGMMIAAAWVLSFILWAPAILFWQFIVGVRTVEDGECYIQF
FSNAAVTFGTAIAAFYLPVIIMTVLYWHISRASKSRIKKDKKEPVANQDPVSPSLVQGRI
VKPNNNNMPSSDDGLEHNKIQNGKAPRDPVTENCVQGEEKESSNDSTSVSAVASNMRDDE
ITQDENTVSTSLGHSKDENSKQTCIRIGTKTPKSDSCTPTNTTVEVVGSSGQNGDEKQNI
VARKIVKMTKQPAKKKPPPSREKKVTRTILAILLAFIITWAPYNVMVLINTFCAPCIPNT
VWTIGYWLCYINSTINPACYALCNATFKKTFKHLLMCHYKNIGATR
|
|
|
|---|
| BDBM50228363 |
|---|
| n/a |
|---|
| Name | BDBM50228363 |
|---|
| Synonyms: | CHEMBL257991 | N-(2-(3-(piperazin-1-ylmethyl)imidazo[2,1-b]thiazol-6-yl)phenyl)quinoxaline-2-carboxamide | SRT-1720 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H23N7OS |
|---|
| Mol. Mass. | 469.561 |
|---|
| SMILES | O=C(Nc1ccccc1-c1cn2c(CN3CCNCC3)csc2n1)c1cnc2ccccc2n1 |
|---|
| Structure | 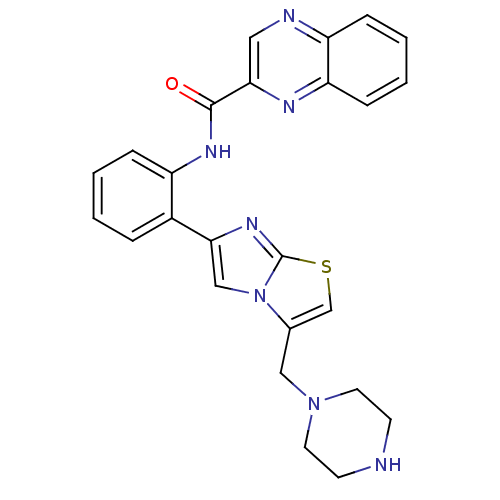
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Vu, CB; Bemis, JE; Disch, JS; Ng, PY; Nunes, JJ; Milne, JC; Carney, DP; Lynch, AV; Smith, JJ; Lavu, S; Lambert, PD; Gagne, DJ; Jirousek, MR; Schenk, S; Olefsky, JM; Perni, RB Discovery of imidazo[1,2-b]thiazole derivatives as novel SIRT1 activators. J Med Chem52:1275-83 (2010) [PubMed] Article
Vu, CB; Bemis, JE; Disch, JS; Ng, PY; Nunes, JJ; Milne, JC; Carney, DP; Lynch, AV; Smith, JJ; Lavu, S; Lambert, PD; Gagne, DJ; Jirousek, MR; Schenk, S; Olefsky, JM; Perni, RB Discovery of imidazo[1,2-b]thiazole derivatives as novel SIRT1 activators. J Med Chem52:1275-83 (2010) [PubMed] Article