

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Serine/threonine-protein kinase mTOR | ||
| Ligand | BDBM50306629 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_611243 (CHEMBL1068272) | ||
| IC50 | 0.3±n/a nM | ||
| Citation |  Curran, KJ; Verheijen, JC; Kaplan, J; Richard, DJ; Toral-Barza, L; Hollander, I; Lucas, J; Ayral-Kaloustian, S; Yu, K; Zask, A Pyrazolopyrimidines as highly potent and selective, ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR): optimization of the 1-substituent. Bioorg Med Chem Lett20:1440-4 (2010) [PubMed] Article Curran, KJ; Verheijen, JC; Kaplan, J; Richard, DJ; Toral-Barza, L; Hollander, I; Lucas, J; Ayral-Kaloustian, S; Yu, K; Zask, A Pyrazolopyrimidines as highly potent and selective, ATP-competitive inhibitors of the mammalian target of rapamycin (mTOR): optimization of the 1-substituent. Bioorg Med Chem Lett20:1440-4 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Serine/threonine-protein kinase mTOR | |||
| Name: | Serine/threonine-protein kinase mTOR | ||
| Synonyms: | FK506-binding protein 12-rapamycin complex-associated protein 1 | FKBP12-rapamycin complex-associated protein | FRAP | FRAP 1 (mTOR) | FRAP1 | FRAP2 | MTOR | MTOR_HUMAN | Mammalian Target of Rapamycin (mTOR) | P42345 | RAFT1 | RAPT1 | Rapamycin and FKBP12 target 1 | Rapamycin target protein | Serine/threonine-protein kinase (mTOR) | mTORC2 | ||
| Type: | Rapamycin target protein | ||
| Mol. Mass.: | 288917.12 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P42345 | ||
| Residue: | 2549 | ||
| Sequence: |
| ||
| BDBM50306629 | |||
| n/a | |||
| Name | BDBM50306629 | ||
| Synonyms: | CHEMBL606621 | cis-1-(4-(4-(8-oxa-3-azabicyclo[3.2.1]octan-3-yl)-1-(4-hydroxycyclohexyl)-1H-pyrazolo[3,4-d]pyrimidin-6-yl)phenyl)-3-methylurea | ||
| Type | Small organic molecule | ||
| Emp. Form. | C25H31N7O3 | ||
| Mol. Mass. | 477.5587 | ||
| SMILES | CNC(=O)Nc1ccc(cc1)-c1nc(N2CC3CCC(C2)O3)c2cnn([C@@H]3CC[C@H](O)CC3)c2n1 |r,wU:26.28,29.32,(3.05,-14.78,;4.38,-15.55,;5.71,-14.77,;5.7,-13.24,;7.04,-15.54,;8.37,-14.77,;8.37,-13.23,;9.7,-12.46,;11.03,-13.23,;11.04,-14.76,;9.71,-15.53,;12.36,-12.45,;12.36,-10.9,;13.7,-10.13,;13.7,-8.58,;12.36,-7.81,;12.36,-6.26,;12.94,-4.86,;14.45,-4.86,;15.04,-6.26,;15.04,-7.81,;13.7,-5.5,;15.04,-10.9,;16.51,-10.42,;17.42,-11.67,;16.52,-12.92,;17,-14.38,;16.19,-15.69,;16.92,-17.04,;18.46,-17.08,;19.19,-18.44,;19.26,-15.77,;18.53,-14.41,;15.04,-12.45,;13.7,-13.22,)| | ||
| Structure | 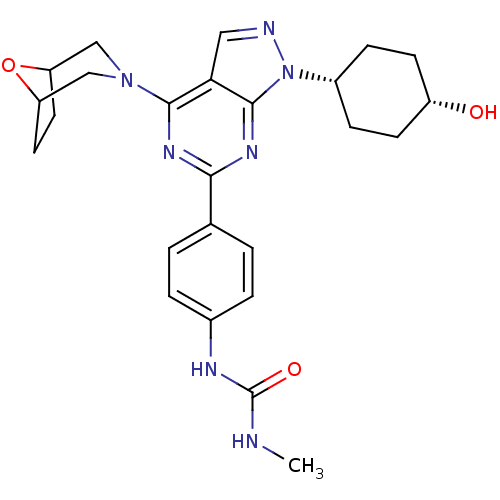
| ||