

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Receptor tyrosine-protein kinase erbB-2 | ||
| Ligand | BDBM50313647 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_614502 (CHEMBL1111475) | ||
| IC50 | 81.0±n/a nM | ||
| Citation |  Morphy, R Selectively nonselective kinase inhibition: striking the right balance. J Med Chem53:1413-37 (2010) [PubMed] Article Morphy, R Selectively nonselective kinase inhibition: striking the right balance. J Med Chem53:1413-37 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Receptor tyrosine-protein kinase erbB-2 | |||
| Name: | Receptor tyrosine-protein kinase erbB-2 | ||
| Synonyms: | 2.7.10.1 | C-erbB-2 | CD_antigen=CD340 | ERBB2 | ERBB2_HUMAN | ErbB-2/ErbB-3 heterodimer | FASN/HER2 | HER-2 Substrate | HER2 | MLN 19 | MLN19 | Metastatic lymph node gene 19 protein | NEU | NGL | Proto-oncogene Neu | Proto-oncogene c-ErbB-2 | Tyrosine kinase-type cell surface receptor HER2 | p185erbB2 | ||
| Type: | n/a | ||
| Mol. Mass.: | 137894.47 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P04626 | ||
| Residue: | 1255 | ||
| Sequence: |
| ||
| BDBM50313647 | |||
| n/a | |||
| Name | BDBM50313647 | ||
| Synonyms: | 3-(2-(2-chlorobenzyl)-1H-benzo[d]imidazol-6-yl)-1-((1r,4r)-4-(1-(3-methoxypropyl)piperidin-4-yl)cyclohexyl)-1H-pyrazolo[3,4-d]pyrimidin-4-amine | CHEMBL2374457 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C34H41ClN8O | ||
| Mol. Mass. | 613.195 | ||
| SMILES | COCCCN1CCC(CC1)[C@H]1CC[C@@H](CC1)n1nc(-c2ccc3nc(Cc4ccccc4Cl)[nH]c3c2)c2c(N)ncnc12 |r,wU:14.18,wD:11.11,(7.5,-14.98,;6.88,-13.57,;7.78,-12.33,;7.16,-10.92,;8.06,-9.67,;7.43,-8.27,;8.34,-7.02,;7.71,-5.61,;6.18,-5.45,;5.28,-6.7,;5.9,-8.11,;5.55,-4.05,;6.46,-2.8,;5.83,-1.39,;4.3,-1.23,;3.4,-2.48,;4.02,-3.89,;3.68,.17,;2.17,.49,;2.01,2.03,;.67,2.8,;.67,4.34,;-.66,5.11,;-1.99,4.34,;-3.46,4.81,;-4.36,3.57,;-5.9,3.57,;-6.67,4.9,;-5.9,6.23,;-6.67,7.57,;-8.21,7.57,;-8.98,6.23,;-8.21,4.9,;-8.98,3.57,;-3.46,2.32,;-1.99,2.8,;-.66,2.03,;3.42,2.65,;3.89,4.12,;2.86,5.26,;5.4,4.44,;6.43,3.29,;5.95,1.83,;4.45,1.51,)| | ||
| Structure | 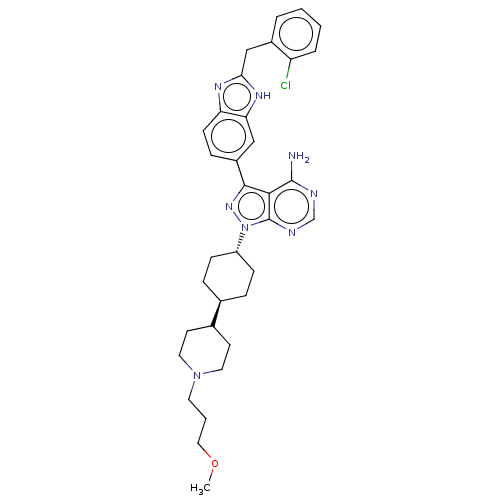
| ||