| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50315595 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_627567 (CHEMBL1113573) |
|---|
| Ki | 38.9±n/a nM |
|---|
| Citation |  Pastorin, G; Federico, S; Paoletta, S; Corradino, M; Cateni, F; Cacciari, B; Klotz, KN; Gao, ZG; Jacobson, KA; Spalluto, G; Moro, S Synthesis and pharmacological characterization of a new series of 5,7-disubstituted-[1,2,4]triazolo[1,5-a][1,3,5]triazine derivatives as adenosine receptor antagonists: A preliminary inspection of ligand-receptor recognition process. Bioorg Med Chem18:2524-36 (2010) [PubMed] Article Pastorin, G; Federico, S; Paoletta, S; Corradino, M; Cateni, F; Cacciari, B; Klotz, KN; Gao, ZG; Jacobson, KA; Spalluto, G; Moro, S Synthesis and pharmacological characterization of a new series of 5,7-disubstituted-[1,2,4]triazolo[1,5-a][1,3,5]triazine derivatives as adenosine receptor antagonists: A preliminary inspection of ligand-receptor recognition process. Bioorg Med Chem18:2524-36 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | AA2AR_RAT | ADENOSINE A2a | Adenosine A2 receptor | Adenosine A2a receptor (A2a) | Adenosine Receptors A2a (A2a) | Adenosine receptor A2a and A3 | Adenosine receptors A2a | Adora2a | Rat striatal adenosine A2a receptor |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 45015.65 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat A2A receptors expressed in CHO cells. |
|---|
| Residue: | 410 |
|---|
| Sequence: | MGSSVYITVELAIAVLAILGNVLVCWAVWINSNLQNVTNFFVVSLAAADIAVGVLAIPFA
ITISTGFCAACHGCLFFACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGVRAKG
IIAICWVLSFAIGLTPMLGWNNCSQKDGNSTKTCGEGRVTCLFEDVVPMNYMVYYNFFAF
VLLPLLLMLAIYLRIFLAARRQLKQMESQPLPGERTRSTLQKEVHAAKSLAIIVGLFALC
WLPLHIINCFTFFCSTCRHAPPWLMYLAIILSHSNSVVNPFIYAYRIREFRQTFRKIIRT
HVLRRQEPFQAGGSSAWALAAHSTEGEQVSLRLNGHPLGVWANGSATHSGRRPNGYTLGL
GGGGSAQGSPRDVELPTQERQEGQEHPGLRGHLVQARVGASSWSSEFAPS
|
|
|
|---|
| BDBM50315595 |
|---|
| n/a |
|---|
| Name | BDBM50315595 |
|---|
| Synonyms: | CHEMBL1089440 | N-(2-(furan-2-yl)-5-phenoxy-[1,2,4]triazolo[1,5-a][1,3,5]triazin-7-yl)hexanamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H15N7O3 |
|---|
| Mol. Mass. | 413.3889 |
|---|
| SMILES | O=C(Nc1ccccc1)Nc1nc(Oc2ccccc2)nc2nc(nn12)-c1ccco1 |
|---|
| Structure | 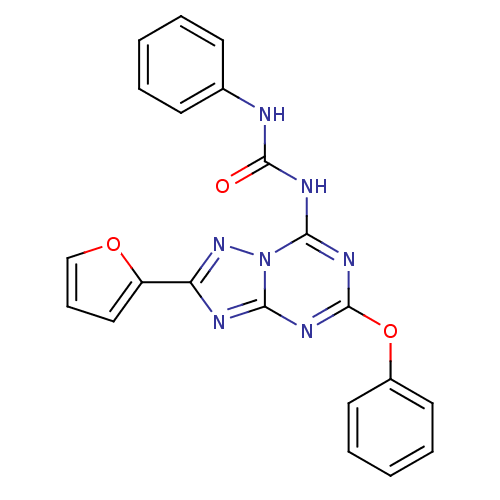
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pastorin, G; Federico, S; Paoletta, S; Corradino, M; Cateni, F; Cacciari, B; Klotz, KN; Gao, ZG; Jacobson, KA; Spalluto, G; Moro, S Synthesis and pharmacological characterization of a new series of 5,7-disubstituted-[1,2,4]triazolo[1,5-a][1,3,5]triazine derivatives as adenosine receptor antagonists: A preliminary inspection of ligand-receptor recognition process. Bioorg Med Chem18:2524-36 (2010) [PubMed] Article
Pastorin, G; Federico, S; Paoletta, S; Corradino, M; Cateni, F; Cacciari, B; Klotz, KN; Gao, ZG; Jacobson, KA; Spalluto, G; Moro, S Synthesis and pharmacological characterization of a new series of 5,7-disubstituted-[1,2,4]triazolo[1,5-a][1,3,5]triazine derivatives as adenosine receptor antagonists: A preliminary inspection of ligand-receptor recognition process. Bioorg Med Chem18:2524-36 (2010) [PubMed] Article