

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform | ||
| Ligand | BDBM50323730 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_646800 (CHEMBL1216941) | ||
| IC50 | 750±n/a nM | ||
| Citation |  Berndt, A; Miller, S; Williams, O; Le, DD; Houseman, BT; Pacold, JI; Gorrec, F; Hon, WC; Liu, Y; Rommel, C; Gaillard, P; Rückle, T; Schwarz, MK; Shokat, KM; Shaw, JP; Williams, RL The p110 delta structure: mechanisms for selectivity and potency of new PI(3)K inhibitors. Nat Chem Biol6:117-24 (2010) [PubMed] Article Berndt, A; Miller, S; Williams, O; Le, DD; Houseman, BT; Pacold, JI; Gorrec, F; Hon, WC; Liu, Y; Rommel, C; Gaillard, P; Rückle, T; Schwarz, MK; Shokat, KM; Shaw, JP; Williams, RL The p110 delta structure: mechanisms for selectivity and potency of new PI(3)K inhibitors. Nat Chem Biol6:117-24 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform | |||
| Name: | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform | ||
| Synonyms: | PI3-kinase p110 subunit beta | PI3-kinase subunit p110-beta | PI3Kbeta | PIK3C1 | PIK3CB | PK3CB_HUMAN | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta (PI3Kbeta) | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform (PI3K beta) | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform (PI3K) | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform (PI3K-beta) | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform (PI3Kbeta) | Phosphatidylinositol 4,5-bisphosphate 3-kinase catalytic subunit beta isoform (PI3Kÿ²) | Phosphatidylinositol-4,5-bisphosphate 3-kinase catalytic subunit beta isoform | Phosphoinositide 3-Kinase (PI3K), beta | Phosphoinositide 3-Kinase (PI3K), beta Chain A | Phosphoinositide-3-kinase (PI3K beta) | PtdIns-3-kinase p110 | ||
| Type: | Enzyme Subunit | ||
| Mol. Mass.: | 122769.00 | ||
| Organism: | Homo sapiens (Human) | ||
| Description: | P42338 | ||
| Residue: | 1070 | ||
| Sequence: |
| ||
| BDBM50323730 | |||
| n/a | |||
| Name | BDBM50323730 | ||
| Synonyms: | 2-((4-amino-3-(3-hydroxyprop-1-ynyl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl)methyl)-5-methyl-3-o-tolylquinazolin-4(3H)-one | 2-{[4-amino-3-(3-hydroxyprop-1-yn-1-yl)-1H-pyrazolo[3,4-d]pyrimidin-1-yl]methyl}-5-methyl-3-(2-methylphenyl)quinazolin-4(3H)-one | CHEMBL1213116 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C25H21N7O2 | ||
| Mol. Mass. | 451.4799 | ||
| SMILES | Cc1ccccc1-n1c(Cn2nc(C#CCO)c3c(N)ncnc23)nc2cccc(C)c2c1=O |(31.57,-15.15,;31.56,-13.61,;32.89,-12.84,;32.88,-11.3,;31.54,-10.53,;30.21,-11.32,;30.22,-12.85,;28.9,-13.62,;28.91,-15.18,;30.25,-15.94,;30.25,-17.49,;29.01,-18.4,;29.49,-19.86,;28.6,-21.11,;27.7,-22.36,;26.81,-23.6,;25.48,-23.68,;31.03,-19.85,;32.06,-20.99,;31.59,-22.46,;33.56,-20.67,;34.03,-19.2,;33,-18.06,;31.51,-18.39,;27.57,-15.96,;26.23,-15.18,;24.89,-15.96,;23.57,-15.19,;23.57,-13.64,;24.89,-12.87,;24.89,-11.33,;26.23,-13.64,;27.56,-12.87,;27.55,-11.33,)| | ||
| Structure | 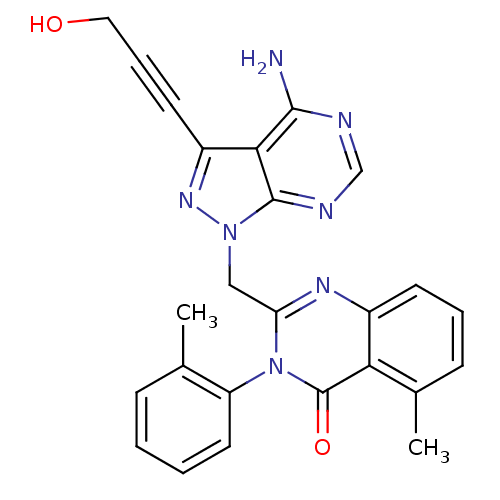
| ||