| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM50001786 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_665659 (CHEMBL1261590) |
|---|
| Ki | 0.2±n/a nM |
|---|
| Citation |  Kristensen, JL; Püschl, A; Jensen, M; Risgaard, R; Christoffersen, CT; Bang-Andersen, B; Balle, T Exploring the neuroleptic substituent in octoclothepin: potential ligands for positron emission tomography with subnanomolar affinity fora(1)-adrenoceptors. J Med Chem53:7021-34 (2010) [PubMed] Article Kristensen, JL; Püschl, A; Jensen, M; Risgaard, R; Christoffersen, CT; Bang-Andersen, B; Balle, T Exploring the neuroleptic substituent in octoclothepin: potential ligands for positron emission tomography with subnanomolar affinity fora(1)-adrenoceptors. J Med Chem53:7021-34 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT-1C | 5-HT-2C | 5-HT1C | 5-HT2C | 5-HT2C-INI | 5-HT2c VGI | 5-HTR2C | 5-hydroxytryptamine receptor 1C | 5-hydroxytryptamine receptor 2C (5-HT-2C) | 5-hydroxytryptamine receptor 2C (5HT-2C) | 5HT-1C | 5HT2C_HUMAN | HTR1C | HTR2C | Serotonin (5-HT3) receptor | Serotonin 2c (5-HT2c) receptor | Serotonin Receptor 2C |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 51836.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28335 |
|---|
| Residue: | 458 |
|---|
| Sequence: | MVNLRNAVHSFLVHLIGLLVWQSDISVSPVAAIVTDIFNTSDGGRFKFPDGVQNWPALSI
VIIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYVW
PLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVWA
ISIGVSVPIPVIGLRDEEKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYCLTIYV
LRRQALMLLHGHTEEPPGLSLDFLKCCKRNTAEEENSANPNQDQNARRRKKKERRPRGTM
QAINNERKASKVLGIVFFVFLIMWCPFFITNILSVLCEKSCNQKLMEKLLNVFVWIGYVC
SGINPLVYTLFNKIYRRAFSNYLRCNYKVEKKPPVRQIPRVAATALSGRELNVNIYRHTN
EPVIEKASDNEPGIEMQVENLELPVNPSSVVSERISSV
|
|
|
|---|
| BDBM50001786 |
|---|
| n/a |
|---|
| Name | BDBM50001786 |
|---|
| Synonyms: | 1-(2-{4-[5-chloro-1-(4-fluorophenyl)-1H-indol-3-yl]piperidin-1-yl}ethyl)imidazolidin-2-one | 1-[2-[4-[5-chloro-1-(4-fluorophenyl)-indol-3-yl]-1-piperidyl]ethyl]imidazolidin-2-one | CHEMBL12713 | Sertindole |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H26ClFN4O |
|---|
| Mol. Mass. | 440.941 |
|---|
| SMILES | Fc1ccc(cc1)-n1cc(C2CCN(CCN3CCNC3=O)CC2)c2cc(Cl)ccc12 |
|---|
| Structure | 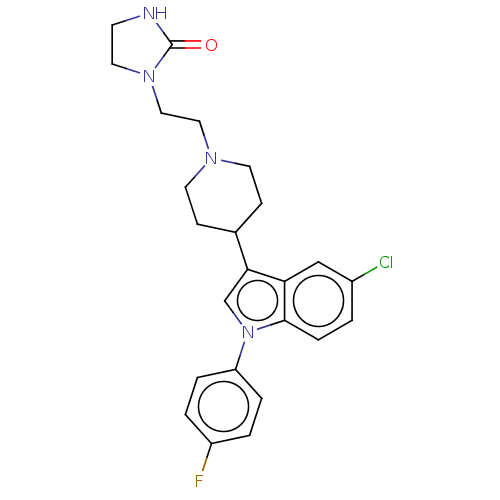
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kristensen, JL; Püschl, A; Jensen, M; Risgaard, R; Christoffersen, CT; Bang-Andersen, B; Balle, T Exploring the neuroleptic substituent in octoclothepin: potential ligands for positron emission tomography with subnanomolar affinity fora(1)-adrenoceptors. J Med Chem53:7021-34 (2010) [PubMed] Article
Kristensen, JL; Püschl, A; Jensen, M; Risgaard, R; Christoffersen, CT; Bang-Andersen, B; Balle, T Exploring the neuroleptic substituent in octoclothepin: potential ligands for positron emission tomography with subnanomolar affinity fora(1)-adrenoceptors. J Med Chem53:7021-34 (2010) [PubMed] Article