| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50329726 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_674334 (CHEMBL1274534) |
|---|
| IC50 | 1100±n/a nM |
|---|
| Citation |  Yang, Z; Fairfax, DJ; Maeng, JH; Masih, L; Usyatinsky, A; Hassler, C; Isaacson, S; Fitzpatrick, K; DeOrazio, RJ; Chen, J; Harding, JP; Isherwood, M; Dobritsa, S; Christensen, KL; Wierschke, JD; Bliss, BI; Peterson, LH; Beer, CM; Cioffi, C; Lynch, M; Rennells, WM; Richards, JJ; Rust, T; Khmelnitsky, YL; Cohen, ML; Manning, DD Discovery of 2-substituted benzoxazole carboxamides as 5-HT3 receptor antagonists. Bioorg Med Chem Lett20:6538-41 (2010) [PubMed] Article Yang, Z; Fairfax, DJ; Maeng, JH; Masih, L; Usyatinsky, A; Hassler, C; Isaacson, S; Fitzpatrick, K; DeOrazio, RJ; Chen, J; Harding, JP; Isherwood, M; Dobritsa, S; Christensen, KL; Wierschke, JD; Bliss, BI; Peterson, LH; Beer, CM; Cioffi, C; Lynch, M; Rennells, WM; Richards, JJ; Rust, T; Khmelnitsky, YL; Cohen, ML; Manning, DD Discovery of 2-substituted benzoxazole carboxamides as 5-HT3 receptor antagonists. Bioorg Med Chem Lett20:6538-41 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50329726 |
|---|
| n/a |
|---|
| Name | BDBM50329726 |
|---|
| Synonyms: | CHEMBL1272017 | endo-2-(2-methoxyphenyl)-N-((1R,5R)-9-methyl-9-azabicyclo[3.3.1]nonan-3-yl)benzo[d]oxazole-4-carboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H27N3O3 |
|---|
| Mol. Mass. | 405.4895 |
|---|
| SMILES | COc1ccccc1-c1nc2c(cccc2o1)C(=O)NC1C[C@H]2CCC[C@H](C1)N2C |r,TLB:29:28:23.24.25:27.20.21| |
|---|
| Structure | 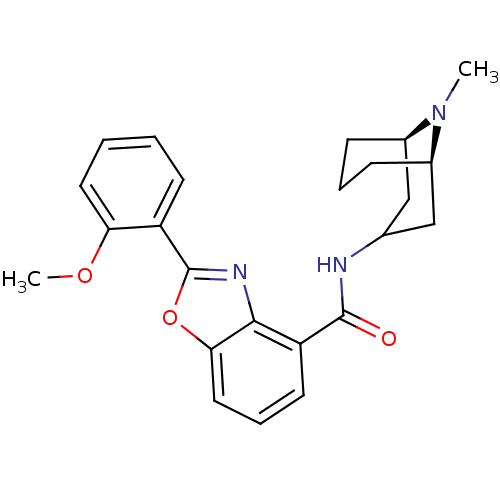
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yang, Z; Fairfax, DJ; Maeng, JH; Masih, L; Usyatinsky, A; Hassler, C; Isaacson, S; Fitzpatrick, K; DeOrazio, RJ; Chen, J; Harding, JP; Isherwood, M; Dobritsa, S; Christensen, KL; Wierschke, JD; Bliss, BI; Peterson, LH; Beer, CM; Cioffi, C; Lynch, M; Rennells, WM; Richards, JJ; Rust, T; Khmelnitsky, YL; Cohen, ML; Manning, DD Discovery of 2-substituted benzoxazole carboxamides as 5-HT3 receptor antagonists. Bioorg Med Chem Lett20:6538-41 (2010) [PubMed] Article
Yang, Z; Fairfax, DJ; Maeng, JH; Masih, L; Usyatinsky, A; Hassler, C; Isaacson, S; Fitzpatrick, K; DeOrazio, RJ; Chen, J; Harding, JP; Isherwood, M; Dobritsa, S; Christensen, KL; Wierschke, JD; Bliss, BI; Peterson, LH; Beer, CM; Cioffi, C; Lynch, M; Rennells, WM; Richards, JJ; Rust, T; Khmelnitsky, YL; Cohen, ML; Manning, DD Discovery of 2-substituted benzoxazole carboxamides as 5-HT3 receptor antagonists. Bioorg Med Chem Lett20:6538-41 (2010) [PubMed] Article