

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
| Reaction Details | |||
|---|---|---|---|
 | Report a problem with these data | ||
| Target | 5-hydroxytryptamine receptor 1A | ||
| Ligand | BDBM50335620 | ||
| Substrate/Competitor | n/a | ||
| Meas. Tech. | ChEMBL_715435 (CHEMBL1664820) | ||
| Ki | 19.9±n/a nM | ||
| Citation |  Herold, F; Chodkowski, A; Izbicki, L; Turlo, J; Dawidowski, M; Kleps, J; Nowak, G; Stachowicz, K; Dybala, M; Siwek, A; Mazurek, AP; Mazurek, A; Plucinski, F Novel 4-aryl-pyrido[1,2-c]pyrimidines with dual SSRI and 5-HT(1A) activity. part 3. Eur J Med Chem46:142-9 (2010) [PubMed] Article Herold, F; Chodkowski, A; Izbicki, L; Turlo, J; Dawidowski, M; Kleps, J; Nowak, G; Stachowicz, K; Dybala, M; Siwek, A; Mazurek, AP; Mazurek, A; Plucinski, F Novel 4-aryl-pyrido[1,2-c]pyrimidines with dual SSRI and 5-HT(1A) activity. part 3. Eur J Med Chem46:142-9 (2010) [PubMed] Article | ||
| More Info.: | Get all data from this article, Assay Method | ||
| 5-hydroxytryptamine receptor 1A | |||
| Name: | 5-hydroxytryptamine receptor 1A | ||
| Synonyms: | 5-HT-1A | 5-HT1 | 5-HT1A | 5-Hydroxytryptamine receptor 1A (5-HT1A) | 5-hydroxytryptamine receptor 1A (5HT1A) | 5HT1A_RAT | 5ht1a | G-21 | Htr1a | Serotonin 1 (5-HT1) receptor | Serotonin 1a (5-HT1a) receptor/Adrenergic receptor alpha-1 | Serotonin receptor 1A | ||
| Type: | G Protein-Coupled Receptor (GPCR) | ||
| Mol. Mass.: | 46445.29 | ||
| Organism: | Rattus norvegicus (rat) | ||
| Description: | Binding assays were performed using rat hippocampal membranes. | ||
| Residue: | 422 | ||
| Sequence: |
| ||
| BDBM50335620 | |||
| n/a | |||
| Name | BDBM50335620 | ||
| Synonyms: | 2-(4-(4-(1H-indol-3-yl)piperidin-1-yl)butyl)-4-(2-methoxyphenyl)-5,6,7,8-tetrahydro-1H-pyrido[1,2-c]pyrimidine-1,3(2H)-dione | CHEMBL1651735 | ||
| Type | Small organic molecule | ||
| Emp. Form. | C32H38N4O3 | ||
| Mol. Mass. | 526.6691 | ||
| SMILES | COc1ccccc1-c1c2CCCCn2c(=O)n(CCCCN2CCC(CC2)c2c[nH]c3ccccc23)c1=O |(.87,-34.6,;-.47,-35.37,;-1.8,-34.61,;-1.81,-33.06,;-3.14,-32.29,;-4.47,-33.06,;-4.47,-34.61,;-3.14,-35.38,;-3.15,-36.91,;-4.48,-37.68,;-5.81,-36.91,;-7.14,-37.67,;-7.14,-39.21,;-5.81,-39.98,;-4.49,-39.21,;-3.16,-39.99,;-3.16,-41.53,;-1.82,-39.22,;-.49,-40,;.85,-39.23,;2.18,-40.01,;3.52,-39.24,;4.85,-40.02,;4.84,-41.56,;6.16,-42.33,;7.5,-41.57,;7.51,-40.03,;6.18,-39.25,;8.83,-42.35,;8.98,-43.88,;10.49,-44.21,;11.26,-42.88,;12.76,-42.58,;13.25,-41.13,;12.23,-39.97,;10.73,-40.28,;10.25,-41.73,;-1.81,-37.68,;-.48,-36.91,)| | ||
| Structure | 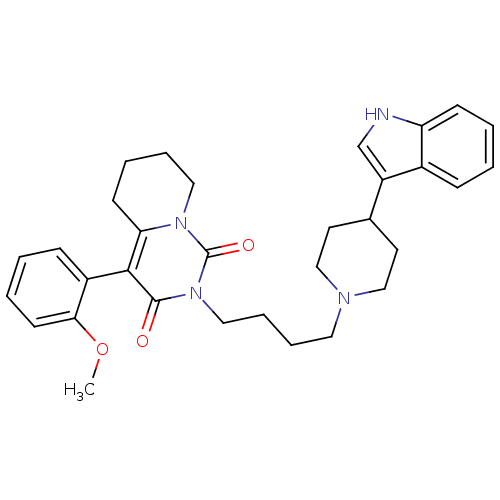
| ||