| Reaction Details |
|---|
 | Report a problem with these data |
| Target | D-alanyl-D-alanine dipeptidase |
|---|
| Ligand | BDBM50335519 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_724225 (CHEMBL1677412) |
|---|
| Ki | 62000±n/a nM |
|---|
| Citation |  Hill, CM; Krause, KM; Lewis, SR; Blais, J; Benton, BM; Mammen, M; Humphrey, PP; Kinana, A; Janc, JW Specificity of induction of the vanA and vanB operons in vancomycin-resistant enterococci by telavancin. Antimicrob Agents Chemother54:2814-8 (2010) [PubMed] Article Hill, CM; Krause, KM; Lewis, SR; Blais, J; Benton, BM; Mammen, M; Humphrey, PP; Kinana, A; Janc, JW Specificity of induction of the vanA and vanB operons in vancomycin-resistant enterococci by telavancin. Antimicrob Agents Chemother54:2814-8 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| D-alanyl-D-alanine dipeptidase |
|---|
| Name: | D-alanyl-D-alanine dipeptidase |
|---|
| Synonyms: | D-Ala-D-Ala dipeptidase | VANX_ENTFC | Vancomycin B-type resistance protein vanX | vanX |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 23377.40 |
|---|
| Organism: | Enterococcus faecium |
|---|
| Description: | ChEMBL_1350288 |
|---|
| Residue: | 202 |
|---|
| Sequence: | MEIGFTFLDEIVHGVRWDAKYATWDNFTGKPVDGYEVNRIVGTYELAESLLKAKELAATQ
GYGLLLWDGYRPKRAVNCFMQWAAQPENNLTKESYYPNIDRTEMISKGYVASKSSHSRGS
AIDLTLYRLDTGELVPMGSRFDFMDERSHHAANGISCNEAQNRRRLRSIMENSGFEAYSL
EWWHYVLRDEPYPNSYFDFPVK
|
|
|
|---|
| BDBM50335519 |
|---|
| n/a |
|---|
| Name | BDBM50335519 |
|---|
| Synonyms: | (S)-3,6-Diamino-hexanoic acid {(3S,9S,12S,15S)-3-((S)-6-hydroxy-2-imino-hexahydro-pyrimidin-4-yl)-9,12-bis-hydroxymethyl-2,5,8,11,14-pentaoxo-6-[1-ureido-meth-(Z)-ylidene]-1,4,7,10,13pentaaza-cyclohexadec-15-yl}-amide | 4-Naphthalen-2-ylmethyl-4,5-dihydro-1H-imidazole | CHEMBL262777 | Chloroorienticin B derivative | Glycopeptide antibiotic | Mannopeptimycin Glycopeptide | Mannopeptimycin glycopeptide analogue | N-demethylvancomycin derivative | Poly ethylene glycol derivative | Tuberactinomycin Analogue | Vancocin | Vancoled | Vancor | Viomycin derivative | vancomycin | vanomycin |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C66H75Cl2N9O24 |
|---|
| Mol. Mass. | 1449.254 |
|---|
| SMILES | CN[C@H](CC(C)C)C(=O)N[C@@H]1[C@H](O)c2ccc(Oc3cc4cc(Oc5ccc(cc5Cl)[C@@H](O)[C@@H]5NC(=O)[C@H](NC(=O)[C@@H]4NC(=O)[C@H](CC(N)=O)NC1=O)c1ccc(O)c(c1)-c1c(O)cc(O)cc1[C@H](NC5=O)C(O)=O)c3O[C@@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1O[C@H]1C[C@](C)(N)[C@H](O)[C@H](C)O1)c(Cl)c2 |r| |
|---|
| Structure | 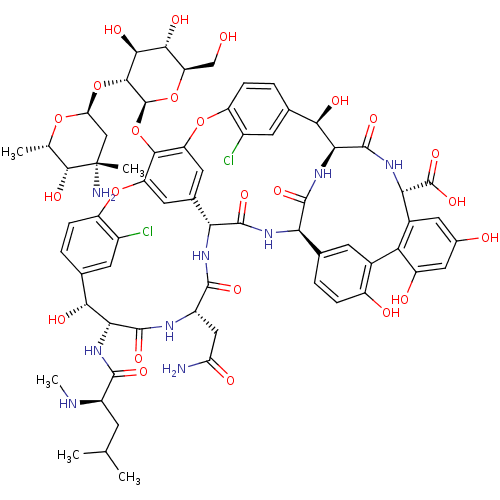
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hill, CM; Krause, KM; Lewis, SR; Blais, J; Benton, BM; Mammen, M; Humphrey, PP; Kinana, A; Janc, JW Specificity of induction of the vanA and vanB operons in vancomycin-resistant enterococci by telavancin. Antimicrob Agents Chemother54:2814-8 (2010) [PubMed] Article
Hill, CM; Krause, KM; Lewis, SR; Blais, J; Benton, BM; Mammen, M; Humphrey, PP; Kinana, A; Janc, JW Specificity of induction of the vanA and vanB operons in vancomycin-resistant enterococci by telavancin. Antimicrob Agents Chemother54:2814-8 (2010) [PubMed] Article