| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C9 |
|---|
| Ligand | BDBM50336978 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_719025 (CHEMBL1679087) |
|---|
| IC50 | >25000±n/a nM |
|---|
| Citation |  Eastwood, P; Esteve, C; González, J; Fonquerna, S; Aiguadé, J; Carranco, Is; Doménech, T; Aparici, Ms; Miralpeix, M; Albertí, J; Córdoba, M; Fernández, R; Pont, Ms; Godessart, Ns; Prats, N; Loza, Ms; Cadavid, Ms; Nueda, A Discovery of LAS101057: A Potent, Selective, and Orally Efficacious A2B Adenosine Receptor Antagonist ACS Med Chem Lett2:213-218 (2011) [PubMed] Article Eastwood, P; Esteve, C; González, J; Fonquerna, S; Aiguadé, J; Carranco, Is; Doménech, T; Aparici, Ms; Miralpeix, M; Albertí, J; Córdoba, M; Fernández, R; Pont, Ms; Godessart, Ns; Prats, N; Loza, Ms; Cadavid, Ms; Nueda, A Discovery of LAS101057: A Potent, Selective, and Orally Efficacious A2B Adenosine Receptor Antagonist ACS Med Chem Lett2:213-218 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C9 |
|---|
| Name: | Cytochrome P450 2C9 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | CP2C9_HUMAN | CYP2C10 | CYP2C9 | CYPIIC9 | Cytochrome P450 2C9 (CYP2C9 ) | Cytochrome P450 2C9 (CYP2C9) | P-450MP | P450 MP-4/MP-8 | P450 PB-1 | S-mephenytoin 4-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55636.33 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P11712 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDSLVVLVLCLSCLLLLSLWRQSSGRGKLPPGPTPLPVIGNILQIGIKDISKSLTNLSKV
YGPVFTLYFGLKPIVVLHGYEAVKEALIDLGEEFSGRGIFPLAERANRGFGIVFSNGKKW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFHKRFDYKDQQFLNLMEKLNENIKILSSPWIQICNNFSPIIDYFPGTHNKLLKNVAFM
KSYILEKVKEHQESMDMNNPQDFIDCFLMKMEKEKHNQPSEFTIESLENTAVDLFGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRSHMPYTDAVVHEVQRYID
LLPTSLPHAVTCDIKFRNYLIPKGTTILISLTSVLHDNKEFPNPEMFDPHHFLDEGGNFK
KSKYFMPFSAGKRICVGEALAGMELFLFLTSILQNFNLKSLVDPKNLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50336978 |
|---|
| n/a |
|---|
| Name | BDBM50336978 |
|---|
| Synonyms: | CHEMBL1672630 | N-(5-(3-chloropyridin-4-yl)-6-(pyridin-2-yl)pyrazin-2-yl)cyclopropanecarboxamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H14FN5O |
|---|
| Mol. Mass. | 335.3351 |
|---|
| SMILES | Fc1cnccc1-c1ncc(NC(=O)C2CC2)nc1-c1ccncc1 |
|---|
| Structure | 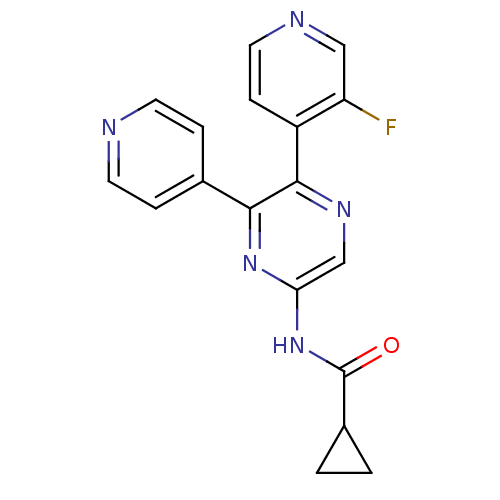
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Eastwood, P; Esteve, C; González, J; Fonquerna, S; Aiguadé, J; Carranco, Is; Doménech, T; Aparici, Ms; Miralpeix, M; Albertí, J; Córdoba, M; Fernández, R; Pont, Ms; Godessart, Ns; Prats, N; Loza, Ms; Cadavid, Ms; Nueda, A Discovery of LAS101057: A Potent, Selective, and Orally Efficacious A2B Adenosine Receptor Antagonist ACS Med Chem Lett2:213-218 (2011) [PubMed] Article
Eastwood, P; Esteve, C; González, J; Fonquerna, S; Aiguadé, J; Carranco, Is; Doménech, T; Aparici, Ms; Miralpeix, M; Albertí, J; Córdoba, M; Fernández, R; Pont, Ms; Godessart, Ns; Prats, N; Loza, Ms; Cadavid, Ms; Nueda, A Discovery of LAS101057: A Potent, Selective, and Orally Efficacious A2B Adenosine Receptor Antagonist ACS Med Chem Lett2:213-218 (2011) [PubMed] Article