| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50341712 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_743387 (CHEMBL1767686) |
|---|
| IC50 | >100000±n/a nM |
|---|
| Citation |  Charrier, JD; Durrant, SJ; Golec, JM; Kay, DP; Knegtel, RM; MacCormick, S; Mortimore, M; O'Donnell, ME; Pinder, JL; Reaper, PM; Rutherford, AP; Wang, PS; Young, SC; Pollard, JR Discovery of potent and selective inhibitors of ataxia telangiectasia mutated and Rad3 related (ATR) protein kinase as potential anticancer agents. J Med Chem54:2320-30 (2011) [PubMed] Article Charrier, JD; Durrant, SJ; Golec, JM; Kay, DP; Knegtel, RM; MacCormick, S; Mortimore, M; O'Donnell, ME; Pinder, JL; Reaper, PM; Rutherford, AP; Wang, PS; Young, SC; Pollard, JR Discovery of potent and selective inhibitors of ataxia telangiectasia mutated and Rad3 related (ATR) protein kinase as potential anticancer agents. J Med Chem54:2320-30 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50341712 |
|---|
| n/a |
|---|
| Name | BDBM50341712 |
|---|
| Synonyms: | 3-amino-N,6-diphenylpyrazine-2-carboxamide | CHEMBL1766759 | US10479784, Compound I-15 | US10961232, Compound I-15 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H14N4O |
|---|
| Mol. Mass. | 290.3193 |
|---|
| SMILES | Nc1ncc(nc1C(=O)Nc1ccccc1)-c1ccccc1 |
|---|
| Structure | 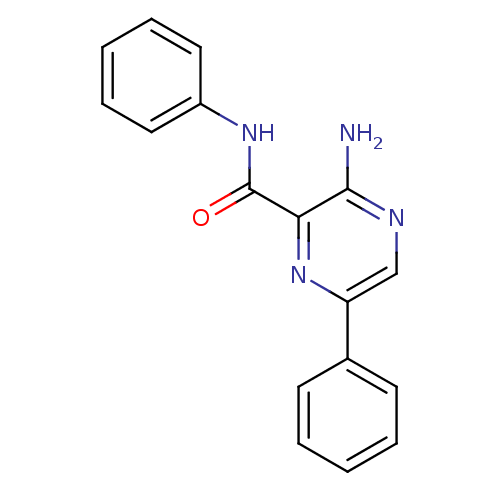
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Charrier, JD; Durrant, SJ; Golec, JM; Kay, DP; Knegtel, RM; MacCormick, S; Mortimore, M; O'Donnell, ME; Pinder, JL; Reaper, PM; Rutherford, AP; Wang, PS; Young, SC; Pollard, JR Discovery of potent and selective inhibitors of ataxia telangiectasia mutated and Rad3 related (ATR) protein kinase as potential anticancer agents. J Med Chem54:2320-30 (2011) [PubMed] Article
Charrier, JD; Durrant, SJ; Golec, JM; Kay, DP; Knegtel, RM; MacCormick, S; Mortimore, M; O'Donnell, ME; Pinder, JL; Reaper, PM; Rutherford, AP; Wang, PS; Young, SC; Pollard, JR Discovery of potent and selective inhibitors of ataxia telangiectasia mutated and Rad3 related (ATR) protein kinase as potential anticancer agents. J Med Chem54:2320-30 (2011) [PubMed] Article