| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2C19 |
|---|
| Ligand | BDBM50342133 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_742782 (CHEMBL1769741) |
|---|
| IC50 | 350±n/a nM |
|---|
| Citation |  Gomaa, MS; Bridgens, CE; Aboraia, AS; Veal, GJ; Redfern, CP; Brancale, A; Armstrong, JL; Simons, C Small molecule inhibitors of retinoic acid 4-hydroxylase (CYP26): synthesis and biological evaluation of imidazole methyl 3-(4-(aryl-2-ylamino)phenyl)propanoates. J Med Chem54:2778-91 (2011) [PubMed] Article Gomaa, MS; Bridgens, CE; Aboraia, AS; Veal, GJ; Redfern, CP; Brancale, A; Armstrong, JL; Simons, C Small molecule inhibitors of retinoic acid 4-hydroxylase (CYP26): synthesis and biological evaluation of imidazole methyl 3-(4-(aryl-2-ylamino)phenyl)propanoates. J Med Chem54:2778-91 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2C19 |
|---|
| Name: | Cytochrome P450 2C19 |
|---|
| Synonyms: | (R)-limonene 6-monooxygenase | (S)-limonene 6-monooxygenase | (S)-limonene 7-monooxygenase | CP2CJ_HUMAN | CYP2C19 | CYPIIC17 | CYPIIC19 | Cytochrome P450 2C19 (CYP2C19) | Cytochrome P450 2C19 [I331V] | Cytochrome P450-11A | Cytochrome P450-254C | Fenbendazole monooxygenase (4'-hydroxylating) | Mephenytoin 4-hydroxylase | P450-11A | P450-254C |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55935.47 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P33261 |
|---|
| Residue: | 490 |
|---|
| Sequence: | MDPFVVLVLCLSCLLLLSIWRQSSGRGKLPPGPTPLPVIGNILQIDIKDVSKSLTNLSKI
YGPVFTLYFGLERMVVLHGYEVVKEALIDLGEEFSGRGHFPLAERANRGFGIVFSNGKRW
KEIRRFSLMTLRNFGMGKRSIEDRVQEEARCLVEELRKTKASPCDPTFILGCAPCNVICS
IIFQKRFDYKDQQFLNLMEKLNENIRIVSTPWIQICNNFPTIIDYFPGTHNKLLKNLAFM
ESDILEKVKEHQESMDINNPRDFIDCFLIKMEKEKQNQQSEFTIENLVITAADLLGAGTE
TTSTTLRYALLLLLKHPEVTAKVQEEIERVIGRNRSPCMQDRGHMPYTDAVVHEVQRYID
LIPTSLPHAVTCDVKFRNYLIPKGTTILTSLTSVLHDNKEFPNPEMFDPRHFLDEGGNFK
KSNYFMPFSAGKRICVGEGLARMELFLFLTFILQNFNLKSLIDPKDLDTTPVVNGFASVP
PFYQLCFIPV
|
|
|
|---|
| BDBM50342133 |
|---|
| n/a |
|---|
| Name | BDBM50342133 |
|---|
| Synonyms: | Benzothiazol-2-yl-[4-((1S,2S)-2-dimethylamino-1-imidazol-1-yl-propyl)-phenyl]-amine | CHEMBL517438 | N-(4-((1R,2R)-2-(dimethylamino)-1-(1H-imidazol-1-yl)propyl)phenyl)benzo[d]thiazol-2-amine | R-116010 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C21H23N5S |
|---|
| Mol. Mass. | 377.506 |
|---|
| SMILES | C[C@H]([C@@H](c1ccc(Nc2nc3ccccc3s2)cc1)n1ccnc1)N(C)C |r| |
|---|
| Structure | 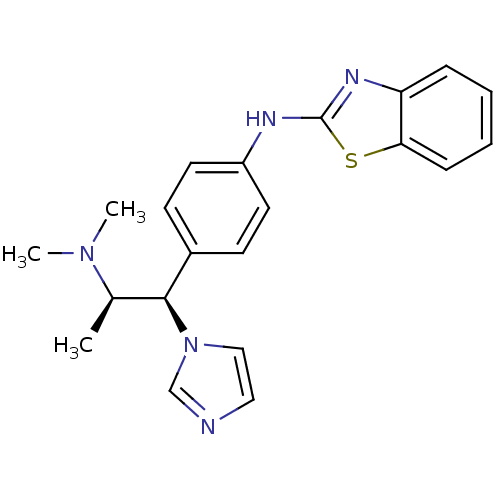
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gomaa, MS; Bridgens, CE; Aboraia, AS; Veal, GJ; Redfern, CP; Brancale, A; Armstrong, JL; Simons, C Small molecule inhibitors of retinoic acid 4-hydroxylase (CYP26): synthesis and biological evaluation of imidazole methyl 3-(4-(aryl-2-ylamino)phenyl)propanoates. J Med Chem54:2778-91 (2011) [PubMed] Article
Gomaa, MS; Bridgens, CE; Aboraia, AS; Veal, GJ; Redfern, CP; Brancale, A; Armstrong, JL; Simons, C Small molecule inhibitors of retinoic acid 4-hydroxylase (CYP26): synthesis and biological evaluation of imidazole methyl 3-(4-(aryl-2-ylamino)phenyl)propanoates. J Med Chem54:2778-91 (2011) [PubMed] Article