| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cystic fibrosis transmembrane conductance regulator |
|---|
| Ligand | BDBM50343998 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_748468 (CHEMBL1780321) |
|---|
| IC50 | 15±n/a nM |
|---|
| Citation |  Renard, BL; Boucherle, B; Maurin, B; Molina, MC; Norez, C; Becq, F; Décout, JL An expeditious access to 5-pyrimidinol derivatives from cyclic methylglyoxal diadducts, formation of argpyrimidines under physiological conditions and discovery of new CFTR inhibitors. Eur J Med Chem46:1935-41 (2011) [PubMed] Article Renard, BL; Boucherle, B; Maurin, B; Molina, MC; Norez, C; Becq, F; Décout, JL An expeditious access to 5-pyrimidinol derivatives from cyclic methylglyoxal diadducts, formation of argpyrimidines under physiological conditions and discovery of new CFTR inhibitors. Eur J Med Chem46:1935-41 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cystic fibrosis transmembrane conductance regulator |
|---|
| Name: | Cystic fibrosis transmembrane conductance regulator |
|---|
| Synonyms: | ABCC7 | ATP-binding cassette sub-family C member 7 | CFTR | CFTR_HUMAN | Channel conductance-controlling ATPase | cAMP-dependent chloride channel |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 168171.30 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | gi_90421313 |
|---|
| Residue: | 1480 |
|---|
| Sequence: | MQRSPLEKASVVSKLFFSWTRPILRKGYRQRLELSDIYQIPSVDSADNLSEKLEREWDRE
LASKKNPKLINALRRCFFWRFMFYGIFLYLGEVTKAVQPLLLGRIIASYDPDNKEERSIA
IYLGIGLCLLFIVRTLLLHPAIFGLHHIGMQMRIAMFSLIYKKTLKLSSRVLDKISIGQL
VSLLSNNLNKFDEGLALAHFVWIAPLQVALLMGLIWELLQASAFCGLGFLIVLALFQAGL
GRMMMKYRDQRAGKISERLVITSEMIENIQSVKAYCWEEAMEKMIENLRQTELKLTRKAA
YVRYFNSSAFFFSGFFVVFLSVLPYALIKGIILRKIFTTISFCIVLRMAVTRQFPWAVQT
WYDSLGAINKIQDFLQKQEYKTLEYNLTTTEVVMENVTAFWEEGFGELFEKAKQNNNNRK
TSNGDDSLFFSNFSLLGTPVLKDINFKIERGQLLAVAGSTGAGKTSLLMVIMGELEPSEG
KIKHSGRISFCSQFSWIMPGTIKENIIFGVSYDEYRYRSVIKACQLEEDISKFAEKDNIV
LGEGGITLSGGQRARISLARAVYKDADLYLLDSPFGYLDVLTEKEIFESCVCKLMANKTR
ILVTSKMEHLKKADKILILHEGSSYFYGTFSELQNLQPDFSSKLMGCDSFDQFSAERRNS
ILTETLHRFSLEGDAPVSWTETKKQSFKQTGEFGEKRKNSILNPINSIRKFSIVQKTPLQ
MNGIEEDSDEPLERRLSLVPDSEQGEAILPRISVISTGPTLQARRRQSVLNLMTHSVNQG
QNIHRKTTASTRKVSLAPQANLTELDIYSRRLSQETGLEISEEINEEDLKECFFDDMESI
PAVTTWNTYLRYITVHKSLIFVLIWCLVIFLAEVAASLVVLWLLGNTPLQDKGNSTHSRN
NSYAVIITSTSSYYVFYIYVGVADTLLAMGFFRGLPLVHTLITVSKILHHKMLHSVLQAP
MSTLNTLKAGGILNRFSKDIAILDDLLPLTIFDFIQLLLIVIGAIAVVAVLQPYIFVATV
PVIVAFIMLRAYFLQTSQQLKQLESEGRSPIFTHLVTSLKGLWTLRAFGRQPYFETLFHK
ALNLHTANWFLYLSTLRWFQMRIEMIFVIFFIAVTFISILTTGEGEGRVGIILTLAMNIM
STLQWAVNSSIDVDSLMRSVSRVFKFIDMPTEGKPTKSTKPYKNGQLSKVMIIENSHVKK
DDIWPSGGQMTVKDLTAKYTEGGNAILENISFSISPGQRVGLLGRTGSGKSTLLSAFLRL
LNTEGEIQIDGVSWDSITLQQWRKAFGVIPQKVFIFSGTFRKNLDPYEQWSDQEIWKVAD
EVGLRSVIEQFPGKLDFVLVDGGCVLSHGHKQLMCLARSVLSKAKILLLDEPSAHLDPVT
YQIIRRTLKQAFADCTVILCEHRIEAMLECQQFLVIEENKVRQYDSIQKLLNERSLFRQA
ISPSDRVKLFPHRNSSKCKSKPQIAALKEETEEEVQDTRL
|
|
|
|---|
| BDBM50343998 |
|---|
| n/a |
|---|
| Name | BDBM50343998 |
|---|
| Synonyms: | (2S,3R,4R)-3,4-dihydroxy-2,4-dimethyl-3,4-dihydro-2H-pyrimido[2,1-a]isoquinolin-1-ium-2-carboxylate | CHEMBL1779953 | CHEMBL1779954 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H16N2O4 |
|---|
| Mol. Mass. | 288.2985 |
|---|
| SMILES | CC1(O)C(O)C(C)(N[c+]2n1ccc1ccccc21)C([O-])=O |
|---|
| Structure | 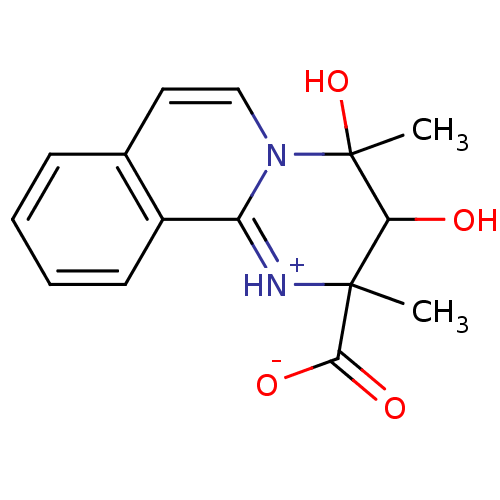
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Renard, BL; Boucherle, B; Maurin, B; Molina, MC; Norez, C; Becq, F; Décout, JL An expeditious access to 5-pyrimidinol derivatives from cyclic methylglyoxal diadducts, formation of argpyrimidines under physiological conditions and discovery of new CFTR inhibitors. Eur J Med Chem46:1935-41 (2011) [PubMed] Article
Renard, BL; Boucherle, B; Maurin, B; Molina, MC; Norez, C; Becq, F; Décout, JL An expeditious access to 5-pyrimidinol derivatives from cyclic methylglyoxal diadducts, formation of argpyrimidines under physiological conditions and discovery of new CFTR inhibitors. Eur J Med Chem46:1935-41 (2011) [PubMed] Article