| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome b-245 heavy chain |
|---|
| Ligand | BDBM50359531 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_792878 (CHEMBL1931063) |
|---|
| Ki | 1760±n/a nM |
|---|
| Citation |  Gaggini, F; Laleu, B; Orchard, M; Fioraso-Cartier, L; Cagnon, L; Houngninou-Molango, S; Gradia, A; Duboux, G; Merlot, C; Heitz, F; Szyndralewiez, C; Page, P Design, synthesis and biological activity of original pyrazolo-pyrido-diazepine, -pyrazine and -oxazine dione derivatives as novel dual Nox4/Nox1 inhibitors. Bioorg Med Chem19:6989-99 (2011) [PubMed] Article Gaggini, F; Laleu, B; Orchard, M; Fioraso-Cartier, L; Cagnon, L; Houngninou-Molango, S; Gradia, A; Duboux, G; Merlot, C; Heitz, F; Szyndralewiez, C; Page, P Design, synthesis and biological activity of original pyrazolo-pyrido-diazepine, -pyrazine and -oxazine dione derivatives as novel dual Nox4/Nox1 inhibitors. Bioorg Med Chem19:6989-99 (2011) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome b-245 heavy chain |
|---|
| Name: | Cytochrome b-245 heavy chain |
|---|
| Synonyms: | CGD91-phox | CY24B_HUMAN | CYBB | Cytochrome b(558) subunit beta | Cytochrome b-245, beta polypeptide [Homo sapiens] | Cytochrome b558 subunit beta | Heme-binding membrane glycoprotein gp91phox | NADPH oxidase 2 | NADPH oxidase 2 (NOX2) | NOX2 | Neutrophil cytochrome b 91 kDa polypeptide | Superoxide-generating NADPH oxidase heavy chain subunit | gp91-1 | gp91-phox | p22 phagocyte B-cytochrome |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 65355.82 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P04839 |
|---|
| Residue: | 570 |
|---|
| Sequence: | MGNWAVNEGLSIFVILVWLGLNVFLFVWYYRVYDIPPKFFYTRKLLGSALALARAPAACL
NFNCMLILLPVCRNLLSFLRGSSACCSTRVRRQLDRNLTFHKMVAWMIALHSAIHTIAHL
FNVEWCVNARVNNSDPYSVALSELGDRQNESYLNFARKRIKNPEGGLYLAVTLLAGITGV
VITLCLILIITSSTKTIRRSYFEVFWYTHHLFVIFFIGLAIHGAERIVRGQTAESLAVHN
ITVCEQKISEWGKIKECPIPQFAGNPPMTWKWIVGPMFLYLCERLVRFWRSQQKVVITKV
VTHPFKTIELQMKKKGFKMEVGQYIFVKCPKVSKLEWHPFTLTSAPEEDFFSIHIRIVGD
WTEGLFNACGCDKQEFQDAWKLPKIAVDGPFGTASEDVFSYEVVMLVGAGIGVTPFASIL
KSVWYKYCNNATNLKLKKIYFYWLCRDTHAFEWFADLLQLLESQMQERNNAGFLSYNIYL
TGWDESQANHFAVHHDEEKDVITGLKQKTLYGRPNWDNEFKTIASQHPNTRIGVFLCGPE
ALAETLSKQSISNSESGPRGVHFIFNKENF
|
|
|
|---|
| BDBM50359531 |
|---|
| n/a |
|---|
| Name | BDBM50359531 |
|---|
| Synonyms: | CHEMBL1927152 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H23ClN4O3 |
|---|
| Mol. Mass. | 450.917 |
|---|
| SMILES | COc1ccccc1CN1CCCn2c(C1)c1c(cc2=O)[nH]n(-c2ccccc2Cl)c1=O |
|---|
| Structure | 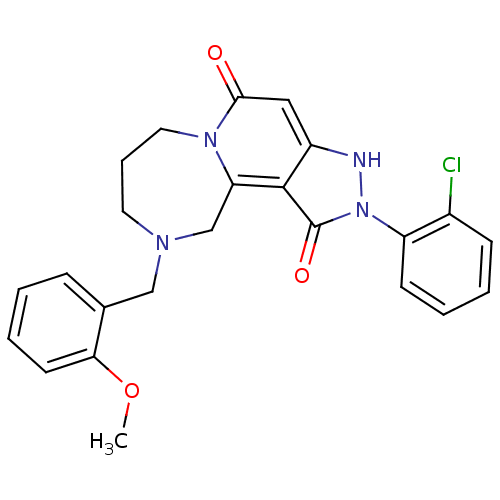
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gaggini, F; Laleu, B; Orchard, M; Fioraso-Cartier, L; Cagnon, L; Houngninou-Molango, S; Gradia, A; Duboux, G; Merlot, C; Heitz, F; Szyndralewiez, C; Page, P Design, synthesis and biological activity of original pyrazolo-pyrido-diazepine, -pyrazine and -oxazine dione derivatives as novel dual Nox4/Nox1 inhibitors. Bioorg Med Chem19:6989-99 (2011) [PubMed] Article
Gaggini, F; Laleu, B; Orchard, M; Fioraso-Cartier, L; Cagnon, L; Houngninou-Molango, S; Gradia, A; Duboux, G; Merlot, C; Heitz, F; Szyndralewiez, C; Page, P Design, synthesis and biological activity of original pyrazolo-pyrido-diazepine, -pyrazine and -oxazine dione derivatives as novel dual Nox4/Nox1 inhibitors. Bioorg Med Chem19:6989-99 (2011) [PubMed] Article