| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sphingosine 1-phosphate receptor 2 |
|---|
| Ligand | BDBM50133422 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_201318 (CHEMBL806168) |
|---|
| EC50 | 2107±n/a nM |
|---|
| Citation |  Clemens, JJ; Davis, MD; Lynch, KR; Macdonald, TL Synthesis of para-alkyl aryl amide analogues of sphingosine-1-phosphate: discovery of potent S1P receptor agonists. Bioorg Med Chem Lett13:3401-4 (2003) [PubMed] Clemens, JJ; Davis, MD; Lynch, KR; Macdonald, TL Synthesis of para-alkyl aryl amide analogues of sphingosine-1-phosphate: discovery of potent S1P receptor agonists. Bioorg Med Chem Lett13:3401-4 (2003) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sphingosine 1-phosphate receptor 2 |
|---|
| Name: | Sphingosine 1-phosphate receptor 2 |
|---|
| Synonyms: | EDG5 | S1P2 | S1PR2 | S1PR2_HUMAN | Sphingosine 1-phosphate receptor | Sphingosine 1-phosphate receptor Edg-5 | Sphingosine-1-phosphate receptor 2 | ndothelial differentiation G-protein coupled receptor 5 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 38883.16 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Membranes isolated from S1P2-transfected CHO cells were used in ligand binding assay. |
|---|
| Residue: | 353 |
|---|
| Sequence: | MGSLYSEYLNPNKVQEHYNYTKETLETQETTSRQVASAFIVILCCAIVVENLLVLIAVAR
NSKFHSAMYLFLGNLAASDLLAGVAFVANTLLSGSVTLRLTPVQWFAREGSAFITLSASV
FSLLAIAIERHVAIAKVKLYGSDKSCRMLLLIGASWLISLVLGGLPILGWNCLGHLEACS
TVLPLYAKHYVLCVVTIFSIILLAIVALYVRIYCVVRSSHADMAAPQTLALLKTVTIVLG
VFIVCWLPAFSILLLDYACPVHSCPILYKAHYFFAVSTLNSLLNPVIYTWRSRDLRREVL
RPLQCWRPGVGVQGRRRGGTPGHHLLPLRSSSSLERGMHMPTSPTFLEGNTVV
|
|
|
|---|
| BDBM50133422 |
|---|
| n/a |
|---|
| Name | BDBM50133422 |
|---|
| Synonyms: | CHEMBL332472 | Phosphoric acid mono-((R)-2-amino-2-tetradecylcarbamoyl-ethyl) ester; TFA |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C17H37N2O5P |
|---|
| Mol. Mass. | 380.4598 |
|---|
| SMILES | CCCCCCCCCCCCCCNC(=O)[C@H](N)COP(O)(O)=O |
|---|
| Structure | 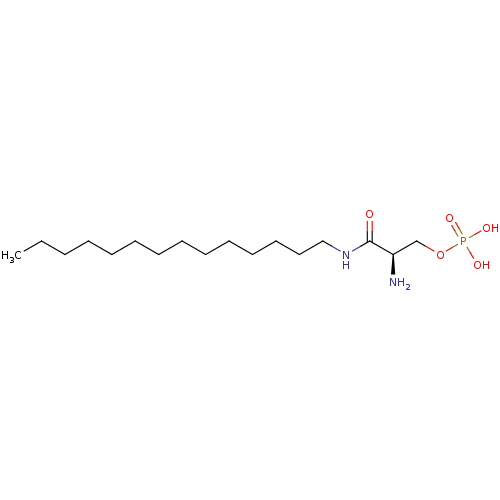
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Clemens, JJ; Davis, MD; Lynch, KR; Macdonald, TL Synthesis of para-alkyl aryl amide analogues of sphingosine-1-phosphate: discovery of potent S1P receptor agonists. Bioorg Med Chem Lett13:3401-4 (2003) [PubMed]
Clemens, JJ; Davis, MD; Lynch, KR; Macdonald, TL Synthesis of para-alkyl aryl amide analogues of sphingosine-1-phosphate: discovery of potent S1P receptor agonists. Bioorg Med Chem Lett13:3401-4 (2003) [PubMed]