| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM21397 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEBML_1137 |
|---|
| Ki | 602±n/a nM |
|---|
| Citation |  Zhuang, ZP; Kung, MP; Kung, HF Synthesis of (R,S)-trans-8-hydroxy-2-[N-n-propyl-N-(3'-iodo-2'-propenyl)amino]tetral in (trans-8-OH-PIPAT): a new 5-HT1A receptor ligand. J Med Chem36:3161-5 (1993) [PubMed] Zhuang, ZP; Kung, MP; Kung, HF Synthesis of (R,S)-trans-8-hydroxy-2-[N-n-propyl-N-(3'-iodo-2'-propenyl)amino]tetral in (trans-8-OH-PIPAT): a new 5-HT1A receptor ligand. J Med Chem36:3161-5 (1993) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1 | 5-HT1A | 5-Hydroxytryptamine receptor 1A (5-HT1A) | 5-hydroxytryptamine receptor 1A (5HT1A) | 5HT1A_RAT | 5ht1a | G-21 | Htr1a | Serotonin 1 (5-HT1) receptor | Serotonin 1a (5-HT1a) receptor/Adrenergic receptor alpha-1 | Serotonin receptor 1A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46445.29 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Binding assays were performed using rat hippocampal membranes. |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVFSFGQGNNTTASQEPFGTGGNVTSISDVTFSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVRKVEKKGAGT
SLGTSSAPPPKKSLNGQPGSGDWRRCAENRAVGTPCTNGAVRQGDDEATLEVIEVHRVGN
SKEHLPLPSESGSNSYAPACLERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPALLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RR
|
|
|
|---|
| BDBM21397 |
|---|
| n/a |
|---|
| Name | BDBM21397 |
|---|
| Synonyms: | 8-[4-(4-fluorophenyl)-4-keto-butyl]-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one | 8-[4-(4-fluorophenyl)-4-oxidanylidene-butyl]-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one | 8-[4-(4-fluorophenyl)-4-oxobutyl]-1-phenyl-1,3,8-triazaspiro[4.5]decan-4-one | CHEMBL267930 | MLS000028615 | SMR000058674 | SPIPERONE | Spiroperidol | US9359372, Spiperone | [3H]-Spiroperidol | cid_5265 |
|---|
| Type | radiolabeled ligand |
|---|
| Emp. Form. | C23H26FN3O2 |
|---|
| Mol. Mass. | 395.4698 |
|---|
| SMILES | Fc1ccc(cc1)C(=O)CCCN1CCC2(CC1)N(CNC2=O)c1ccccc1 |
|---|
| Structure | 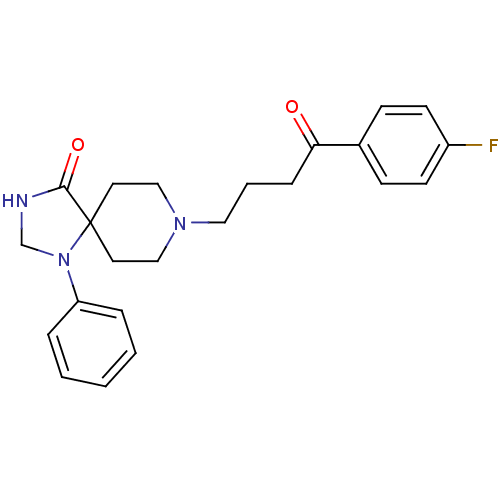
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zhuang, ZP; Kung, MP; Kung, HF Synthesis of (R,S)-trans-8-hydroxy-2-[N-n-propyl-N-(3'-iodo-2'-propenyl)amino]tetral in (trans-8-OH-PIPAT): a new 5-HT1A receptor ligand. J Med Chem36:3161-5 (1993) [PubMed]
Zhuang, ZP; Kung, MP; Kung, HF Synthesis of (R,S)-trans-8-hydroxy-2-[N-n-propyl-N-(3'-iodo-2'-propenyl)amino]tetral in (trans-8-OH-PIPAT): a new 5-HT1A receptor ligand. J Med Chem36:3161-5 (1993) [PubMed]