| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1A adrenergic receptor |
|---|
| Ligand | BDBM50005127 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_32974 (CHEMBL644689) |
|---|
| IC50 | 1200±n/a nM |
|---|
| Citation |  Podona, T; Guardiola-Lemaître, B; Caignard, DH; Adam, G; Pfeiffer, B; Renard, P; Guillaumet, G 3,4-Dihydro-3-amino-2H-1-benzopyran derivatives as 5-HT1A receptor ligands and potential anxiolytic agents. 1. Synthesis and structure--activity relationship studies. J Med Chem37:1779-93 (1994) [PubMed] Podona, T; Guardiola-Lemaître, B; Caignard, DH; Adam, G; Pfeiffer, B; Renard, P; Guillaumet, G 3,4-Dihydro-3-amino-2H-1-benzopyran derivatives as 5-HT1A receptor ligands and potential anxiolytic agents. 1. Synthesis and structure--activity relationship studies. J Med Chem37:1779-93 (1994) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1A adrenergic receptor |
|---|
| Name: | Alpha-1A adrenergic receptor |
|---|
| Synonyms: | ADA1A_BOVIN | ADRA1A | ADRA1C | Alpha 1A-adrenoceptor | Alpha 1A-adrenoreceptor | Alpha-1C adrenergic receptor | Cerebral cortex alpha adrenergic receptor | adrenergic Alpha1 |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 51492.08 |
|---|
| Organism: | CALF |
|---|
| Description: | adrenergic Alpha1 0 CALF |
|---|
| Residue: | 466 |
|---|
| Sequence: | MVFLSGNASDSSNCTHPPPPVNISKAILLGVILGGLILFGVLGNILVILSVACHRHLHSV
THYYIVNLAVADLLLTSTVLPFSAIFEILGYWAFGRVFCNVWAAVDVLCCTASIMGLCII
SIDRYIGVSYPLRYPTIVTQKRGLMALLCVWALSLVISIGPLFGWRQPAPEDETICQINE
EPGYVLFSALGSFYVPLTIILVMYCRVYVVAKRESRGLKSGLKTDKSDSEQVTLRIHRKN
AQVGGSGVTSAKNKTHFSVRLLKFSREKKAAKTLGIVVGCFVLCWLPFFLVMPIGSFFPD
FRPSETVFKIAFWLGYLNSCINPIIYPCSSQEFKKAFQNVLRIQCLRRKQSSKHTLGYTL
HAPSHVLEGQHKDLVRIPVGSAETFYKISKTDGVCEWKIFSSLPRGSARMAVARDPSACT
TARVRSKSFLQVCCCLGPSTPSHGENHQIPTIKIHTISLSENGEEV
|
|
|
|---|
| BDBM50005127 |
|---|
| n/a |
|---|
| Name | BDBM50005127 |
|---|
| Synonyms: | 1,1-Dioxo-2-[4-(4-pyrimidin-2-yl-piperazin-1-yl)-butyl]-1,2-dihydro-1lambda*6*-benzo[d]isothiazol-3-one | 1,1-Dioxo-2-[4-(4-pyrimidin-2-yl-piperazin-1-yl)-butyl]-1,2-dihydro-1lambda*6*-benzo[d]isothiazol-3-one (Ipsapirone) | 1,1-Dioxo-2-[4-(4-pyrimidin-2-yl-piperazin-1-yl)-butyl]-1,2-dihydro-1lambda*6*-benzo[d]isothiazol-3-one(Ipsapirone) | CHEMBL8412 | IPSAPIRONE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H23N5O3S |
|---|
| Mol. Mass. | 401.483 |
|---|
| SMILES | O=C1N(CCCCN2CCN(CC2)c2ncccn2)S(=O)(=O)c2ccccc12 |
|---|
| Structure | 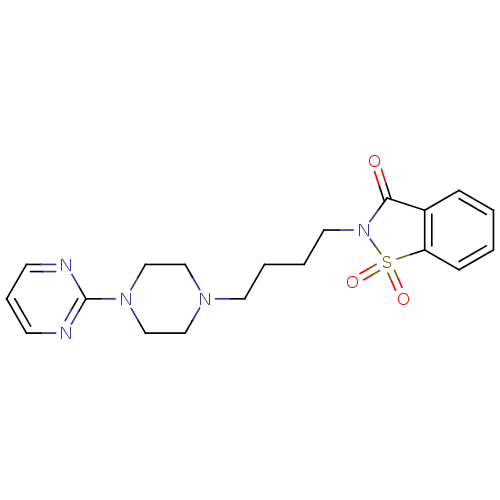
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Podona, T; Guardiola-Lemaître, B; Caignard, DH; Adam, G; Pfeiffer, B; Renard, P; Guillaumet, G 3,4-Dihydro-3-amino-2H-1-benzopyran derivatives as 5-HT1A receptor ligands and potential anxiolytic agents. 1. Synthesis and structure--activity relationship studies. J Med Chem37:1779-93 (1994) [PubMed]
Podona, T; Guardiola-Lemaître, B; Caignard, DH; Adam, G; Pfeiffer, B; Renard, P; Guillaumet, G 3,4-Dihydro-3-amino-2H-1-benzopyran derivatives as 5-HT1A receptor ligands and potential anxiolytic agents. 1. Synthesis and structure--activity relationship studies. J Med Chem37:1779-93 (1994) [PubMed]