| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 3-oxo-5-alpha-steroid 4-dehydrogenase 1 |
|---|
| Ligand | BDBM50043604 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_204756 (CHEMBL882394) |
|---|
| IC50 | >1000±n/a nM |
|---|
| Citation |  Bakshi, RK; Rasmusson, GH; Patel, GF; Mosley, RT; Chang, B; Ellsworth, K; Harris, GS; Tolman, RL 4-Aza-3-oxo-5 alpha-androst-1-ene-17 beta-N-aryl-carboxamides as dual inhibitors of human type 1 and type 2 steroid 5 alpha-reductases. Dramatic effect of N-aryl substituents on type 1 and type 2 5 alpha-reductase inhibitory potency. J Med Chem38:3189-92 (1995) [PubMed] Bakshi, RK; Rasmusson, GH; Patel, GF; Mosley, RT; Chang, B; Ellsworth, K; Harris, GS; Tolman, RL 4-Aza-3-oxo-5 alpha-androst-1-ene-17 beta-N-aryl-carboxamides as dual inhibitors of human type 1 and type 2 steroid 5 alpha-reductases. Dramatic effect of N-aryl substituents on type 1 and type 2 5 alpha-reductase inhibitory potency. J Med Chem38:3189-92 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 3-oxo-5-alpha-steroid 4-dehydrogenase 1 |
|---|
| Name: | 3-oxo-5-alpha-steroid 4-dehydrogenase 1 |
|---|
| Synonyms: | 3-oxo-5-alpha-steroid 4-dehydrogenase 1 | 5α-Reductase 1 (5α-R1) | S5A1_HUMAN | S5AR | SR type 1 | SRD5A1 | Steroid 5-alpha-reductase | Steroid 5-alpha-reductase 1 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 29472.80 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P18405 |
|---|
| Residue: | 259 |
|---|
| Sequence: | MATATGVAEERLLAALAYLQCAVGCAVFARNRQTNSVYGRHALPSHRLRVPARAAWVVQE
LPSLALPLYQYASESAPRLRSAPNCILLAMFLVHYGHRCLIYPFLMRGGKPMPLLACTMA
IMFCTCNGYLQSRYLSHCAVYADDWVTDPRFLIGFGLWLTGMLINIHSDHILRNLRKPGD
TGYKIPRGGLFEYVTAANYFGEIMEWCGYALASWSVQGAAFAFFTFCFLSGRAKEHHEWY
LRKFEEYPKFRKIIIPFLF
|
|
|
|---|
| BDBM50043604 |
|---|
| n/a |
|---|
| Name | BDBM50043604 |
|---|
| Synonyms: | (8S,9S,10R,13S,14S,17S)-17-(tert-butylcarbamoyl)-10,13-dimethyl-2,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthrene-3-carboxylic acid | 17-tert-Butylcarbamoyl-10,13-dimethyl-2,7,8,9,10,11,12,13,14,15,16,17-dodecahydro-1H-cyclopenta[a]phenanthrene-3-carboxylic acid | CHEMBL138225 | Epristeride |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H37NO3 |
|---|
| Mol. Mass. | 399.5662 |
|---|
| SMILES | CC(C)(C)NC(=O)[C@H]1CC[C@H]2[C@@H]3CC=C4C=C(CC[C@]4(C)[C@H]3CC[C@]12C)C(O)=O |c:15,t:13| |
|---|
| Structure | 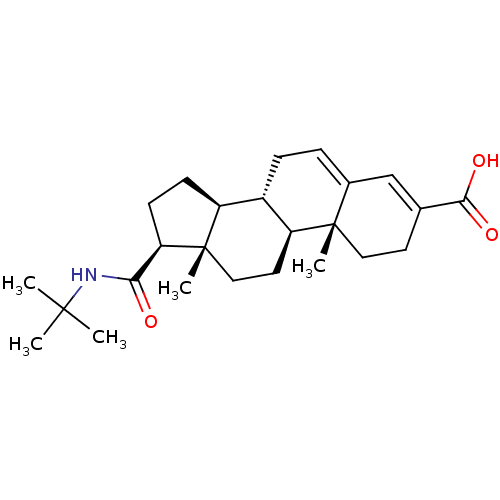
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Bakshi, RK; Rasmusson, GH; Patel, GF; Mosley, RT; Chang, B; Ellsworth, K; Harris, GS; Tolman, RL 4-Aza-3-oxo-5 alpha-androst-1-ene-17 beta-N-aryl-carboxamides as dual inhibitors of human type 1 and type 2 steroid 5 alpha-reductases. Dramatic effect of N-aryl substituents on type 1 and type 2 5 alpha-reductase inhibitory potency. J Med Chem38:3189-92 (1995) [PubMed]
Bakshi, RK; Rasmusson, GH; Patel, GF; Mosley, RT; Chang, B; Ellsworth, K; Harris, GS; Tolman, RL 4-Aza-3-oxo-5 alpha-androst-1-ene-17 beta-N-aryl-carboxamides as dual inhibitors of human type 1 and type 2 steroid 5 alpha-reductases. Dramatic effect of N-aryl substituents on type 1 and type 2 5 alpha-reductase inhibitory potency. J Med Chem38:3189-92 (1995) [PubMed]