| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 3-oxo-5-alpha-steroid 4-dehydrogenase 2 |
|---|
| Ligand | BDBM50034134 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_204737 (CHEMBL805430) |
|---|
| IC50 | >100±n/a nM |
|---|
| Citation |  Li, X; Singh, SM; Labrie, F Synthesis and in vitro activity of 17 beta-(N-alkyl/arylformamido)- and 17 beta-[(N-alkyl/aryl)alkyl/arylamido]-4-methyl-4-aza-3-oxo-5 alpha-androstan-3-ones as inhibitors of human 5 alpha-reductases and antagonists of the androgen receptor. J Med Chem38:1158-73 (1995) [PubMed] Li, X; Singh, SM; Labrie, F Synthesis and in vitro activity of 17 beta-(N-alkyl/arylformamido)- and 17 beta-[(N-alkyl/aryl)alkyl/arylamido]-4-methyl-4-aza-3-oxo-5 alpha-androstan-3-ones as inhibitors of human 5 alpha-reductases and antagonists of the androgen receptor. J Med Chem38:1158-73 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 3-oxo-5-alpha-steroid 4-dehydrogenase 2 |
|---|
| Name: | 3-oxo-5-alpha-steroid 4-dehydrogenase 2 |
|---|
| Synonyms: | 3-oxo-5-alpha-steroid 4-dehydrogenase 2 | 5 alpha-SR2 | 5α-Reductase 2 (5α-R2) | S5A2_HUMAN | SR type 2 | SRD5A2 | Steroid 5-alpha-reductase | Steroid 5-alpha-reductase 2 | Type II 5-alpha reductase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 28406.59 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P31213 |
|---|
| Residue: | 254 |
|---|
| Sequence: | MQVQCQQSPVLAGSATLVALGALALYVAKPSGYGKHTESLKPAATRLPARAAWFLQELPS
FAVPAGILARQPLSLFGPPGTVLLGLFCLHYFHRTFVYSLLNRGRPYPAILILRGTAFCT
GNGVLQGYYLIYCAEYPDGWYTDIRFSLGVFLFILGMGINIHSDYILRQLRKPGEISYRI
PQGGLFTYVSGANFLGEIIEWIGYALATWSLPALAFAFFSLCFLGLRAFHHHRFYLKMFE
DYPKSRKALIPFIF
|
|
|
|---|
| BDBM50034134 |
|---|
| n/a |
|---|
| Name | BDBM50034134 |
|---|
| Synonyms: | CHEMBL17222 | N-(3-Methyl-butyl)-N-((4aR,6aS,7S,11aR)-1,4a,6a-trimethyl-2-oxo-hexadecahydro-indeno[5,4-f]quinolin-7-yl)-formamide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H42N2O2 |
|---|
| Mol. Mass. | 402.6132 |
|---|
| SMILES | CC(C)CCN(C=O)[C@H]1CCC2C3CC[C@H]4N(C)C(=O)CC[C@]4(C)C3CC[C@]12C |
|---|
| Structure | 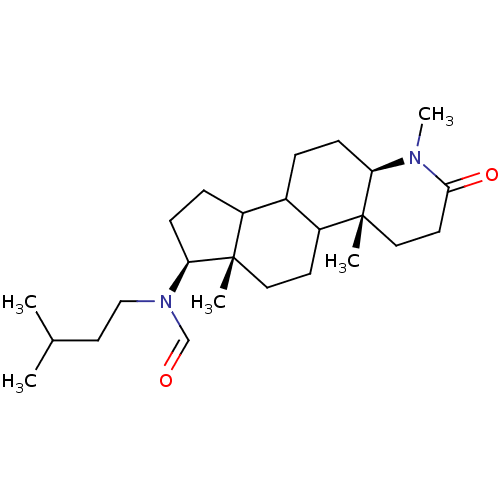
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Li, X; Singh, SM; Labrie, F Synthesis and in vitro activity of 17 beta-(N-alkyl/arylformamido)- and 17 beta-[(N-alkyl/aryl)alkyl/arylamido]-4-methyl-4-aza-3-oxo-5 alpha-androstan-3-ones as inhibitors of human 5 alpha-reductases and antagonists of the androgen receptor. J Med Chem38:1158-73 (1995) [PubMed]
Li, X; Singh, SM; Labrie, F Synthesis and in vitro activity of 17 beta-(N-alkyl/arylformamido)- and 17 beta-[(N-alkyl/aryl)alkyl/arylamido]-4-methyl-4-aza-3-oxo-5 alpha-androstan-3-ones as inhibitors of human 5 alpha-reductases and antagonists of the androgen receptor. J Med Chem38:1158-73 (1995) [PubMed]