| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Progesterone receptor |
|---|
| Ligand | BDBM50366558 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_29970 (CHEMBL642162) |
|---|
| IC50 | 3.2±n/a nM |
|---|
| Citation |  Hamann, LG; Farmer, LJ; Johnson, MG; Bender, SL; Mais, DE; Wang, MW; Crombie, D; Goldman, ME; Jones, TK Synthesis and biological activity of novel nonsteroidal progesterone receptor antagonists based on cyclocymopol monomethyl ether. J Med Chem39:1778-89 (1996) [PubMed] Article Hamann, LG; Farmer, LJ; Johnson, MG; Bender, SL; Mais, DE; Wang, MW; Crombie, D; Goldman, ME; Jones, TK Synthesis and biological activity of novel nonsteroidal progesterone receptor antagonists based on cyclocymopol monomethyl ether. J Med Chem39:1778-89 (1996) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Progesterone receptor |
|---|
| Name: | Progesterone receptor |
|---|
| Synonyms: | NR3C3 | Nuclear receptor subfamily 3 group C member 3 | PGR | PR | PRGR_HUMAN | progesterone |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 98979.52 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P06401 |
|---|
| Residue: | 933 |
|---|
| Sequence: | MTELKAKGPRAPHVAGGPPSPEVGSPLLCRPAAGPFPGSQTSDTLPEVSAIPISLDGLLF
PRPCQGQDPSDEKTQDQQSLSDVEGAYSRAEATRGAGGSSSSPPEKDSGLLDSVLDTLLA
PSGPGQSQPSPPACEVTSSWCLFGPELPEDPPAAPATQRVLSPLMSRSGCKVGDSSGTAA
AHKVLPRGLSPARQLLLPASESPHWSGAPVKPSPQAAAVEVEEEDGSESEESAGPLLKGK
PRALGGAAAGGGAAAVPPGAAAGGVALVPKEDSRFSAPRVALVEQDAPMAPGRSPLATTV
MDFIHVPILPLNHALLAARTRQLLEDESYDGGAGAASAFAPPRSSPCASSTPVAVGDFPD
CAYPPDAEPKDDAYPLYSDFQPPALKIKEEEEGAEASARSPRSYLVAGANPAAFPDFPLG
PPPPLPPRATPSRPGEAAVTAAPASASVSSASSSGSTLECILYKAEGAPPQQGPFAPPPC
KAPGASGCLLPRDGLPSTSASAAAAGAAPALYPALGLNGLPQLGYQAAVLKEGLPQVYPP
YLNYLRPDSEASQSPQYSFESLPQKICLICGDEASGCHYGVLTCGSCKVFFKRAMEGQHN
YLCAGRNDCIVDKIRRKNCPACRLRKCCQAGMVLGGRKFKKFNKVRVVRALDAVALPQPV
GVPNESQALSQRFTFSPGQDIQLIPPLINLLMSIEPDVIYAGHDNTKPDTSSSLLTSLNQ
LGERQLLSVVKWSKSLPGFRNLHIDDQITLIQYSWMSLMVFGLGWRSYKHVSGQMLYFAP
DLILNEQRMKESSFYSLCLTMWQIPQEFVKLQVSQEEFLCMKVLLLLNTIPLEGLRSQTQ
FEEMRSSYIRELIKAIGLRQKGVVSSSQRFYQLTKLLDNLHDLVKQLHLYCLNTFIQSRA
LSVEFPEMMSEVIAAQLPKILAGMVKPLLFHKK
|
|
|
|---|
| BDBM50366558 |
|---|
| n/a |
|---|
| Name | BDBM50366558 |
|---|
| Synonyms: | ONAPRISTONE | ZK-98299 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C29H39NO3 |
|---|
| Mol. Mass. | 449.6249 |
|---|
| SMILES | CN(C)c1ccc(cc1)[C@H]1C[C@]2(C)[C@@H](CC[C@]2(O)CCCO)[C@@H]2CCC3=CC(=O)CCC3=C12 |t:27,34| |
|---|
| Structure | 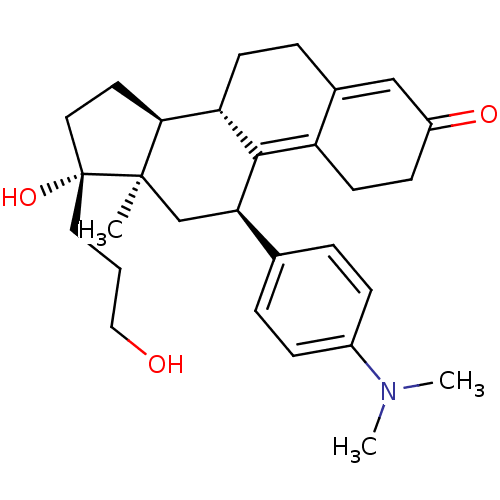
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hamann, LG; Farmer, LJ; Johnson, MG; Bender, SL; Mais, DE; Wang, MW; Crombie, D; Goldman, ME; Jones, TK Synthesis and biological activity of novel nonsteroidal progesterone receptor antagonists based on cyclocymopol monomethyl ether. J Med Chem39:1778-89 (1996) [PubMed] Article
Hamann, LG; Farmer, LJ; Johnson, MG; Bender, SL; Mais, DE; Wang, MW; Crombie, D; Goldman, ME; Jones, TK Synthesis and biological activity of novel nonsteroidal progesterone receptor antagonists based on cyclocymopol monomethyl ether. J Med Chem39:1778-89 (1996) [PubMed] Article