| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Inosine-5'-monophosphate dehydrogenase 2 |
|---|
| Ligand | BDBM50421763 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_89934 (CHEMBL699560) |
|---|
| IC50 | 90±n/a nM |
|---|
| Citation |  Lesiak, K; Watanabe, KA; Majumdar, A; Seidman, M; Vanderveen, K; Goldstein, BM; Pankiewicz, KW Synthesis of nonhydrolyzable analogues of thiazole-4-carboxamide and benzamide adenine dinucleotide containing fluorine atom at the C2' of adenine nucleoside: induction of K562 differentiation and inosine monophosphate dehydrogenase inhibitory activity. J Med Chem40:2533-8 (1997) [PubMed] Article Lesiak, K; Watanabe, KA; Majumdar, A; Seidman, M; Vanderveen, K; Goldstein, BM; Pankiewicz, KW Synthesis of nonhydrolyzable analogues of thiazole-4-carboxamide and benzamide adenine dinucleotide containing fluorine atom at the C2' of adenine nucleoside: induction of K562 differentiation and inosine monophosphate dehydrogenase inhibitory activity. J Med Chem40:2533-8 (1997) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Inosine-5'-monophosphate dehydrogenase 2 |
|---|
| Name: | Inosine-5'-monophosphate dehydrogenase 2 |
|---|
| Synonyms: | IMDH2_HUMAN | IMP dehydrogenase 2 | IMPD 2 | IMPD2 | IMPDH-II | IMPDH2 | Inosine 5'-monophosphate dehydrogenase II (IMPDH II) | Inosine Monophosphate Dehydrogenase Type 2 (IMPDH2) | Inosine-5 -monophosphate dehydrogenase 2 | Inosine-5'-monophosphate dehydrogenase (IMPDH) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 55806.87 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | Recombinant IMPDH2 expressed in E. coli. |
|---|
| Residue: | 514 |
|---|
| Sequence: | MADYLISGGTSYVPDDGLTAQQLFNCGDGLTYNDFLILPGYIDFTADQVDLTSALTKKIT
LKTPLVSSPMDTVTEAGMAIAMALTGGIGFIHHNCTPEFQANEVRKVKKYEQGFITDPVV
LSPKDRVRDVFEAKARHGFCGIPITDTGRMGSRLVGIISSRDIDFLKEEEHDCFLEEIMT
KREDLVVAPAGITLKEANEILQRSKKGKLPIVNEDDELVAIIARTDLKKNRDYPLASKDA
KKQLLCGAAIGTHEDDKYRLDLLAQAGVDVVVLDSSQGNSIFQINMIKYIKDKYPNLQVI
GGNVVTAAQAKNLIDAGVDALRVGMGSGSICITQEVLACGRPQATAVYKVSEYARRFGVP
VIADGGIQNVGHIAKALALGASTVMMGSLLAATTEAPGEYFFSDGIRLKKYRGMGSLDAM
DKHLSSQNRYFSEADKIKVAQGVSGAVQDKGSIHKFVPYLIAGIQHSCQDIGAKSLTQVR
AMMYSGELKFEKRTSSAQVEGGVHSLHSYEKRLF
|
|
|
|---|
| BDBM50421763 |
|---|
| n/a |
|---|
| Name | BDBM50421763 |
|---|
| Synonyms: | CHEMBL2364562 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H25N7O14P2S |
|---|
| Mol. Mass. | 669.453 |
|---|
| SMILES | NC(=O)c1csc(n1)[C@H]1O[C@@H](COP(O)(=O)OP(O)(=O)OC[C@H]2O[C@H]([C@H](O)[C@@H]2O)n2cnc3c(N)ncnc23)[C@H](O)[C@@H]1O |r| |
|---|
| Structure | 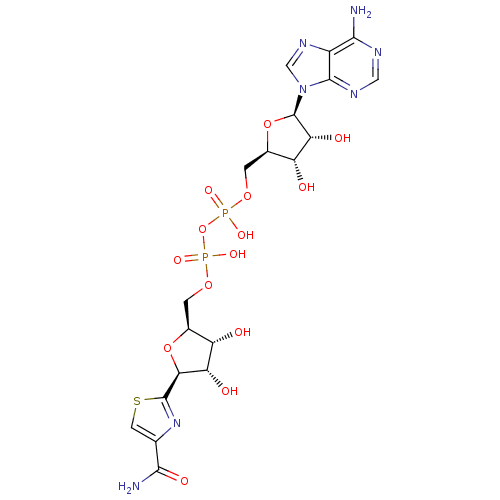
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Lesiak, K; Watanabe, KA; Majumdar, A; Seidman, M; Vanderveen, K; Goldstein, BM; Pankiewicz, KW Synthesis of nonhydrolyzable analogues of thiazole-4-carboxamide and benzamide adenine dinucleotide containing fluorine atom at the C2' of adenine nucleoside: induction of K562 differentiation and inosine monophosphate dehydrogenase inhibitory activity. J Med Chem40:2533-8 (1997) [PubMed] Article
Lesiak, K; Watanabe, KA; Majumdar, A; Seidman, M; Vanderveen, K; Goldstein, BM; Pankiewicz, KW Synthesis of nonhydrolyzable analogues of thiazole-4-carboxamide and benzamide adenine dinucleotide containing fluorine atom at the C2' of adenine nucleoside: induction of K562 differentiation and inosine monophosphate dehydrogenase inhibitory activity. J Med Chem40:2533-8 (1997) [PubMed] Article