| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50066109 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_34340 (CHEMBL649153) |
|---|
| Ki | 0.67±n/a nM |
|---|
| Citation |  Chern, JW; Tao, PL; Wang, KC; Gutcait, A; Liu, SW; Yen, MH; Chien, SL; Rong, JK Studies on quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides. 8.1, 2 synthesis and pharmacological evaluation of tricyclic fused quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides as potential alpha1-adrenoceptor antagonists. J Med Chem41:3128-41 (1998) [PubMed] Article Chern, JW; Tao, PL; Wang, KC; Gutcait, A; Liu, SW; Yen, MH; Chien, SL; Rong, JK Studies on quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides. 8.1, 2 synthesis and pharmacological evaluation of tricyclic fused quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides as potential alpha1-adrenoceptor antagonists. J Med Chem41:3128-41 (1998) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_RAT | Adra1b | Alpha 1B-adrenoceptor | Alpha 1B-adrenoreceptor | Alpha adrenergic receptor 1A and 1B | Alpha-1 Adrenergic Receptor | Alpha-1Adrenoceptor | Alpha-1B adrenergic receptor | Alpha-1B adrenoreceptor | adrenergic Alpha1B |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 56606.71 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Receptor binding assays were performed using rat cortical membranes. |
|---|
| Residue: | 515 |
|---|
| Sequence: | MNPDLDTGHNTSAPAHWGELKDDNFTGPNQTSSNSTLPQLDVTRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAIADLLLSFTVLPFSATLEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFM
RILGCQCRGGRRRRRRRRLGACAYTYRPWTRGGSLERSQSRKDSLDDSGSCMSGTQRTLP
SASPSPGYLGRGTQPPVELCAFPEWKPGALLSLPEPPGRRGRLDSGPLFTFKLLGDPESP
GTEGDTSNGGCDTTTDLANGQPGFKSNMPLAPGHF
|
|
|
|---|
| BDBM50066109 |
|---|
| n/a |
|---|
| Name | BDBM50066109 |
|---|
| Synonyms: | 3-[4-(2-Methoxy-phenyl)-piperazin-1-ylmethyl]-2,6-dihydro-3H-imidazo[1,2-c]quinazolin-5-one | CHEMBL106737 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C22H25N5O2 |
|---|
| Mol. Mass. | 391.4662 |
|---|
| SMILES | COc1ccccc1N1CCN(CC2CN=C3N2C(=O)Nc2ccccc32)CC1 |c:16| |
|---|
| Structure | 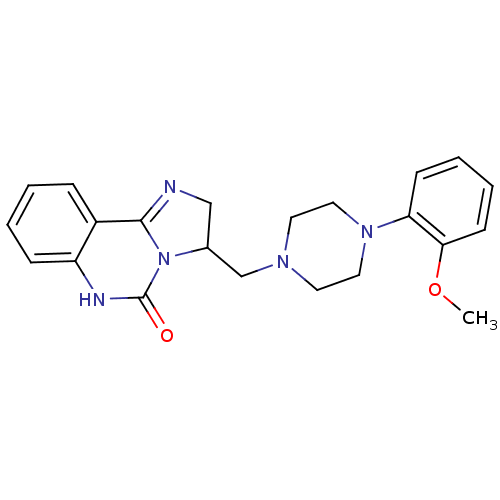
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Chern, JW; Tao, PL; Wang, KC; Gutcait, A; Liu, SW; Yen, MH; Chien, SL; Rong, JK Studies on quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides. 8.1, 2 synthesis and pharmacological evaluation of tricyclic fused quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides as potential alpha1-adrenoceptor antagonists. J Med Chem41:3128-41 (1998) [PubMed] Article
Chern, JW; Tao, PL; Wang, KC; Gutcait, A; Liu, SW; Yen, MH; Chien, SL; Rong, JK Studies on quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides. 8.1, 2 synthesis and pharmacological evaluation of tricyclic fused quinazolines and 1,2,4-benzothiadiazine 1,1-dioxides as potential alpha1-adrenoceptor antagonists. J Med Chem41:3128-41 (1998) [PubMed] Article