| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Alpha-1B adrenergic receptor |
|---|
| Ligand | BDBM50082803 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_34457 (CHEMBL651989) |
|---|
| Ki | 139±n/a nM |
|---|
| Citation |  Nagarathnam, D; Miao, SW; Lagu, B; Chiu, G; Fang, J; Murali Dhar, TG; Zhang, J; Tyagarajan, S; Marzabadi, MR; Zhang, F; Wong, WC; Sun, W; Tian, D; Wetzel, JM; Forray, C; Chang, RS; Broten, TP; Ransom, RW; Schorn, TW; Chen, TB; O'Malley, S; Kling, P; Schneck, K; Bendesky, R; Harrell, CM Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 1. Structure-activity relationship in dihydropyrimidinones. J Med Chem42:4764-77 (1999) [PubMed] Nagarathnam, D; Miao, SW; Lagu, B; Chiu, G; Fang, J; Murali Dhar, TG; Zhang, J; Tyagarajan, S; Marzabadi, MR; Zhang, F; Wong, WC; Sun, W; Tian, D; Wetzel, JM; Forray, C; Chang, RS; Broten, TP; Ransom, RW; Schorn, TW; Chen, TB; O'Malley, S; Kling, P; Schneck, K; Bendesky, R; Harrell, CM Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 1. Structure-activity relationship in dihydropyrimidinones. J Med Chem42:4764-77 (1999) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Alpha-1B adrenergic receptor |
|---|
| Name: | Alpha-1B adrenergic receptor |
|---|
| Synonyms: | ADA1B_HUMAN | ADRA1B | Adrenergic alpha1B | Adrenergic receptor | Adrenergic receptor alpha | Alpha 1B-adrenoceptor | Alpha 1B-adrenoreceptor | Alpha-1B adrenergic receptor |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56862.13 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P35368 |
|---|
| Residue: | 520 |
|---|
| Sequence: | MNPDLDTGHNTSAPAHWGELKNANFTGPNQTSSNSTLPQLDITRAISVGLVLGAFILFAI
VGNILVILSVACNRHLRTPTNYFIVNLAMADLLLSFTVLPFSAALEVLGYWVLGRIFCDI
WAAVDVLCCTASILSLCAISIDRYIGVRYSLQYPTLVTRRKAILALLSVWVLSTVISIGP
LLGWKEPAPNDDKECGVTEEPFYALFSSLGSFYIPLAVILVMYCRVYIVAKRTTKNLEAG
VMKEMSNSKELTLRIHSKNFHEDTLSSTKAKGHNPRSSIAVKLFKFSREKKAAKTLGIVV
GMFILCWLPFFIALPLGSLFSTLKPPDAVFKVVFWLGYFNSCLNPIIYPCSSKEFKRAFV
RILGCQCRGRGRRRRRRRRRLGGCAYTYRPWTRGGSLERSQSRKDSLDDSGSCLSGSQRT
LPSASPSPGYLGRGAPPPVELCAFPEWKAPGALLSLPAPEPPGRRGRHDSGPLFTFKLLT
EPESPGTDGGASNGGCEAAADVANGQPGFKSNMPLAPGQF
|
|
|
|---|
| BDBM50082803 |
|---|
| n/a |
|---|
| Name | BDBM50082803 |
|---|
| Synonyms: | 1-(3-{[4-Ethyl-5-methylcarbamoyl-6-(4-nitro-phenyl)-2-oxo-3,6-dihydro-2H-pyrimidine-1-carbonyl]-amino}-propyl)-4-phenyl-piperidine-4-carboxylic acid methyl ester | CHEMBL145752 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H38N6O7 |
|---|
| Mol. Mass. | 606.6694 |
|---|
| SMILES | CCC1=C(C(N(C(=O)NCCCN2CCC(CC2)(C(=O)OC)c2ccccc2)C(=O)N1)c1ccc(cc1)[N+]([O-])=O)C(=O)NC |t:2| |
|---|
| Structure | 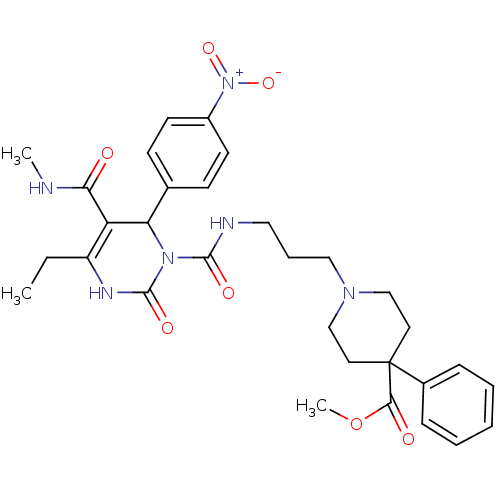
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Nagarathnam, D; Miao, SW; Lagu, B; Chiu, G; Fang, J; Murali Dhar, TG; Zhang, J; Tyagarajan, S; Marzabadi, MR; Zhang, F; Wong, WC; Sun, W; Tian, D; Wetzel, JM; Forray, C; Chang, RS; Broten, TP; Ransom, RW; Schorn, TW; Chen, TB; O'Malley, S; Kling, P; Schneck, K; Bendesky, R; Harrell, CM Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 1. Structure-activity relationship in dihydropyrimidinones. J Med Chem42:4764-77 (1999) [PubMed]
Nagarathnam, D; Miao, SW; Lagu, B; Chiu, G; Fang, J; Murali Dhar, TG; Zhang, J; Tyagarajan, S; Marzabadi, MR; Zhang, F; Wong, WC; Sun, W; Tian, D; Wetzel, JM; Forray, C; Chang, RS; Broten, TP; Ransom, RW; Schorn, TW; Chen, TB; O'Malley, S; Kling, P; Schneck, K; Bendesky, R; Harrell, CM Design and synthesis of novel alpha(1)(a) adrenoceptor-selective antagonists. 1. Structure-activity relationship in dihydropyrimidinones. J Med Chem42:4764-77 (1999) [PubMed]