| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prostaglandin G/H synthase 1 |
|---|
| Ligand | BDBM50094534 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_159260 (CHEMBL764087) |
|---|
| IC50 | 100±n/a nM |
|---|
| Citation |  Portevin, B; Tordjman, C; Pastoureau, P; Bonnet, J; De Nanteuil G, na 1,3-Diaryl-4,5,6,7-tetrahydro-2H-isoindole derivatives: a new series of potent and selective COX-2 inhibitors in which a sulfonyl group is not a structural requisite. J Med Chem43:4582-93 (2001) [PubMed] Portevin, B; Tordjman, C; Pastoureau, P; Bonnet, J; De Nanteuil G, na 1,3-Diaryl-4,5,6,7-tetrahydro-2H-isoindole derivatives: a new series of potent and selective COX-2 inhibitors in which a sulfonyl group is not a structural requisite. J Med Chem43:4582-93 (2001) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prostaglandin G/H synthase 1 |
|---|
| Name: | Prostaglandin G/H synthase 1 |
|---|
| Synonyms: | Cox-1 | Cox1 | Cyclooxygenase-1 | PGH synthase 1 | PGH1_MOUSE | PGHS-1 | PHS 1 | Prostaglandin G/H synthase (cyclooxygenase) | Prostaglandin H2 synthase 1 | Prostaglandin-endoperoxide synthase 1 | Ptgs1 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 69044.61 |
|---|
| Organism: | Mus musculus |
|---|
| Description: | ChEMBL_10575 |
|---|
| Residue: | 602 |
|---|
| Sequence: | MSRRSLSLWFPLLLLLLLPPTPSVLLADPGVPSPVNPCCYYPCQNQGVCVRFGLDNYQCD
CTRTGYSGPNCTIPEIWTWLRNSLRPSPSFTHFLLTHGYWLWEFVNATFIREVLMRLVLT
VRSNLIPSPPTYNSAHDYISWESFSNVSYYTRILPSVPKDCPTPMGTKGKKQLPDVQLLA
QQLLLRREFIPAPQGTNILFAFFAQHFTHQFFKTSGKMGPGFTKALGHGVDLGHIYGDNL
ERQYHLRLFKDGKLKYQVLDGEVYPPSVEQASVLMRYPPGVPPERQMAVGQEVFGLLPGL
MLFSTIWLREHNRVCDLLKEEHPTWDDEQLFQTTRLILIGETIKIVIEEYVQHLSGYFLQ
LKFDPELLFRAQFQYRNRIAMEFNHLYHWHPLMPNSFQVGSQEYSYEQFLFNTSMLVDYG
VEALVDAFSRQRAGRIGGGRNFDYHVLHVAVDVIKESREMRLQPFNEYRKRFGLKPYTSF
QELTGEKEMAAELEELYGDIDALEFYPGLLLEKCQPNSIFGESMIEMGAPFSLKGLLGNP
ICSPEYWKPSTFGGDVGFNLVNTASLKKLVCLNTKTCPYVSFRVPDYPGDDGSVLVRRST
EL
|
|
|
|---|
| BDBM50094534 |
|---|
| n/a |
|---|
| Name | BDBM50094534 |
|---|
| Synonyms: | 1,3-Bis-(4-fluoro-phenyl)-4,5,6,7-tetrahydro-2H-isoindole | 1,3-bis(4-fluorophenyl)-4,5,6,7-tetrahydro-2H-isoindole | CHEMBL441663 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H17F2N |
|---|
| Mol. Mass. | 309.3525 |
|---|
| SMILES | Fc1ccc(cc1)-c1[nH]c(c2CCCCc12)-c1ccc(F)cc1 |
|---|
| Structure | 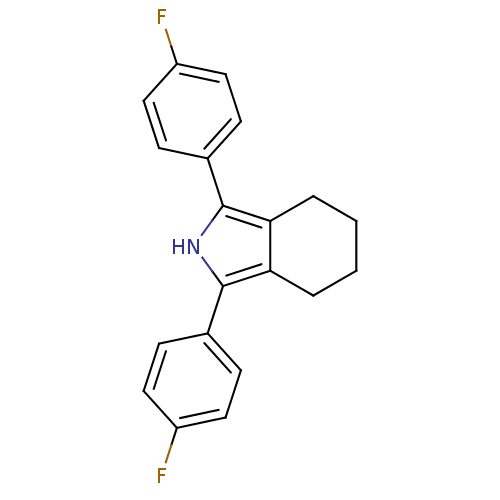
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Portevin, B; Tordjman, C; Pastoureau, P; Bonnet, J; De Nanteuil G, na 1,3-Diaryl-4,5,6,7-tetrahydro-2H-isoindole derivatives: a new series of potent and selective COX-2 inhibitors in which a sulfonyl group is not a structural requisite. J Med Chem43:4582-93 (2001) [PubMed]
Portevin, B; Tordjman, C; Pastoureau, P; Bonnet, J; De Nanteuil G, na 1,3-Diaryl-4,5,6,7-tetrahydro-2H-isoindole derivatives: a new series of potent and selective COX-2 inhibitors in which a sulfonyl group is not a structural requisite. J Med Chem43:4582-93 (2001) [PubMed]