| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50072775 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_46459 (CHEMBL657906) |
|---|
| Ki | 5.2±n/a nM |
|---|
| Citation |  Ooms, F; Wouters, J; Oscari, O; Happaerts, T; Bouchard, G; Carrupt, PA; Testa, B; Lambert, DM Exploration of the pharmacophore of 3-alkyl-5-arylimidazolidinediones as new CB(1) cannabinoid receptor ligands and potential antagonists: synthesis, lipophilicity, affinity, and molecular modeling. J Med Chem45:1748-56 (2002) [PubMed] Ooms, F; Wouters, J; Oscari, O; Happaerts, T; Bouchard, G; Carrupt, PA; Testa, B; Lambert, DM Exploration of the pharmacophore of 3-alkyl-5-arylimidazolidinediones as new CB(1) cannabinoid receptor ligands and potential antagonists: synthesis, lipophilicity, affinity, and molecular modeling. J Med Chem45:1748-56 (2002) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50072775 |
|---|
| n/a |
|---|
| Name | BDBM50072775 |
|---|
| Synonyms: | 2-((1R,2R,5R)-5-hydroxy-2-(3-hydroxypropyl)cyclohexyl)-5-(2-methyloctan-2-yl)phenol | 5-(1,1-Dimethyl-heptyl)-2-[(1R,2R,5R)-5-hydroxy-2-(3-hydroxy-propyl)-cyclohexyl]-phenol | 5-(1,1-Dimethyl-heptyl)-2-[(4R,6R)-5-hydroxy-2-((R)-3-hydroxy-propyl)-cyclohexyl]-phenol | CHEMBL48552 | CP-55940 | CP-55940 [(-)-AC],XIV | CP55,940 | CP55940 | ChEMBL_75020 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C24H40O3 |
|---|
| Mol. Mass. | 376.5726 |
|---|
| SMILES | CCCCCCC(C)(C)c1ccc([C@@H]2C[C@H](O)CC[C@H]2CCCO)c(O)c1 |r| |
|---|
| Structure | 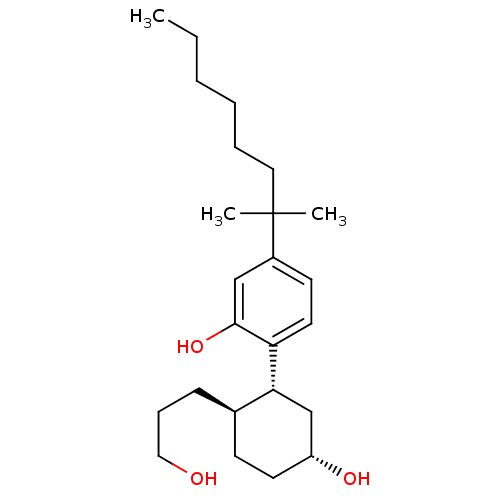
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Ooms, F; Wouters, J; Oscari, O; Happaerts, T; Bouchard, G; Carrupt, PA; Testa, B; Lambert, DM Exploration of the pharmacophore of 3-alkyl-5-arylimidazolidinediones as new CB(1) cannabinoid receptor ligands and potential antagonists: synthesis, lipophilicity, affinity, and molecular modeling. J Med Chem45:1748-56 (2002) [PubMed]
Ooms, F; Wouters, J; Oscari, O; Happaerts, T; Bouchard, G; Carrupt, PA; Testa, B; Lambert, DM Exploration of the pharmacophore of 3-alkyl-5-arylimidazolidinediones as new CB(1) cannabinoid receptor ligands and potential antagonists: synthesis, lipophilicity, affinity, and molecular modeling. J Med Chem45:1748-56 (2002) [PubMed]