| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Sodium-dependent dopamine transporter |
|---|
| Ligand | BDBM50128231 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_61993 (CHEMBL670598) |
|---|
| IC50 | 173±n/a nM |
|---|
| Citation |  Kolhatkar, RB; Ghorai, SK; George, C; Reith, ME; Dutta, AK Interaction of cis-(6-benzhydrylpiperidin-3-yl)benzylamine analogues with monoamine transporters: structure-activity relationship study of structurally constrained 3,6-disubstituted piperidine analogues of (2,2-diphenylethyl)-[1-(4-fluorobenzyl)piperidin-4-ylmethyl]amine. J Med Chem46:2205-15 (2003) [PubMed] Article Kolhatkar, RB; Ghorai, SK; George, C; Reith, ME; Dutta, AK Interaction of cis-(6-benzhydrylpiperidin-3-yl)benzylamine analogues with monoamine transporters: structure-activity relationship study of structurally constrained 3,6-disubstituted piperidine analogues of (2,2-diphenylethyl)-[1-(4-fluorobenzyl)piperidin-4-ylmethyl]amine. J Med Chem46:2205-15 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Sodium-dependent dopamine transporter |
|---|
| Name: | Sodium-dependent dopamine transporter |
|---|
| Synonyms: | DA transporter | Monoamine transporters; Norepininephrine & dopamine | SC6A3_RAT | Slc6a3 | Sodium-dependent dopamine transporter | Sodium-dependent dopamine transporter (DAT) |
|---|
| Type: | Multi-pass membrane protein |
|---|
| Mol. Mass.: | 68749.45 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | P23977 |
|---|
| Residue: | 619 |
|---|
| Sequence: | MSKSKCSVGPMSSVVAPAKESNAVGPREVELILVKEQNGVQLTNSTLINPPQTPVEAQER
ETWSKKIDFLLSVIGFAVDLANVWRFPYLCYKNGGGAFLVPYLLFMVIAGMPLFYMELAL
GQFNREGAAGVWKICPVLKGVGFTVILISFYVGFFYNVIIAWALHYFFSSFTMDLPWIHC
NNTWNSPNCSDAHASNSSDGLGLNDTFGTTPAAEYFERGVLHLHQSRGIDDLGPPRWQLT
ACLVLVIVLLYFSLWKGVKTSGKVVWITATMPYVVLTALLLRGVTLPGAMDGIRAYLSVD
FYRLCEASVWIDAATQVCFSLGVGFGVLIAFSSYNKFTNNCYRDAIITTSINSLTSFSSG
FVVFSFLGYMAQKHNVPIRDVATDGPGLIFIIYPEAIATLPLSSAWAAVFFLMLLTLGID
SAMGGMESVITGLVDEFQLLHRHRELFTLGIVLATFLLSLFCVTNGGIYVFTLLDHFAAG
TSILFGVLIEAIGVAWFYGVQQFSDDIKQMTGQRPNLYWRLCWKLVSPCFLLYVVVVSIV
TFRPPHYGAYIFPDWANALGWIIATSSMAMVPIYATYKFCSLPGSFREKLAYAITPEKDH
QLVDRGEVRQFTLRHWLLL
|
|
|
|---|
| BDBM50128231 |
|---|
| n/a |
|---|
| Name | BDBM50128231 |
|---|
| Synonyms: | (6-Benzhydryl-piperidin-3-yl)-[2-(4-fluoro-phenyl)-ethyl]-amine; dihydrochloride | CHEMBL544288 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H29FN2 |
|---|
| Mol. Mass. | 388.5203 |
|---|
| SMILES | Fc1ccc(CCNC2CCC(NC2)C(c2ccccc2)c2ccccc2)cc1 |
|---|
| Structure | 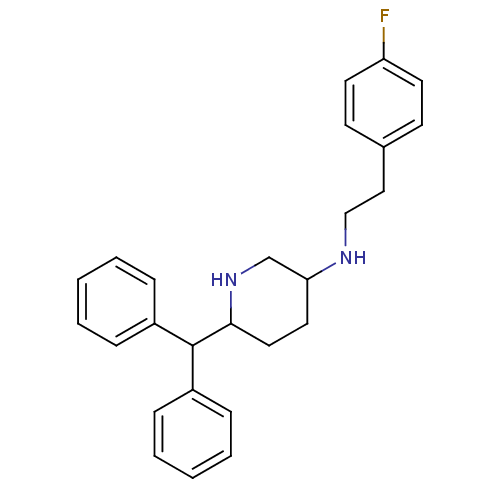
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kolhatkar, RB; Ghorai, SK; George, C; Reith, ME; Dutta, AK Interaction of cis-(6-benzhydrylpiperidin-3-yl)benzylamine analogues with monoamine transporters: structure-activity relationship study of structurally constrained 3,6-disubstituted piperidine analogues of (2,2-diphenylethyl)-[1-(4-fluorobenzyl)piperidin-4-ylmethyl]amine. J Med Chem46:2205-15 (2003) [PubMed] Article
Kolhatkar, RB; Ghorai, SK; George, C; Reith, ME; Dutta, AK Interaction of cis-(6-benzhydrylpiperidin-3-yl)benzylamine analogues with monoamine transporters: structure-activity relationship study of structurally constrained 3,6-disubstituted piperidine analogues of (2,2-diphenylethyl)-[1-(4-fluorobenzyl)piperidin-4-ylmethyl]amine. J Med Chem46:2205-15 (2003) [PubMed] Article