| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 4 |
|---|
| Ligand | BDBM50005833 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_3275 (CHEMBL619076) |
|---|
| Ki | 21±n/a nM |
|---|
| Citation |  Hirokawa, Y; Fujiwara, I; Suzuki, K; Harada, H; Yoshikawa, T; Yoshida, N; Kato, S Synthesis and structure-affinity relationships of novel N-(1-ethyl-4-methylhexahydro-1,4-diazepin-6-yl)pyridine-3-carboxamides with potent serotonin 5-HT3 and dopamine D2 receptor antagonistic activity. J Med Chem46:702-15 (2003) [PubMed] Article Hirokawa, Y; Fujiwara, I; Suzuki, K; Harada, H; Yoshikawa, T; Yoshida, N; Kato, S Synthesis and structure-affinity relationships of novel N-(1-ethyl-4-methylhexahydro-1,4-diazepin-6-yl)pyridine-3-carboxamides with potent serotonin 5-HT3 and dopamine D2 receptor antagonistic activity. J Med Chem46:702-15 (2003) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 4 |
|---|
| Name: | 5-hydroxytryptamine receptor 4 |
|---|
| Synonyms: | 5-HT4 | 5-hydroxytryptamine receptor 4 | 5HT4R_CAVPO | HTR4 | Serotonin 4 (5-HT4) receptor |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 43735.29 |
|---|
| Organism: | GUINEA PIG |
|---|
| Description: | 5-HT4 HTR4 GUINEA PIG::O70528 |
|---|
| Residue: | 388 |
|---|
| Sequence: | MDKLDANVSSKEGFGSVEKVVLLTFLSAVILMAILGNLLVMVAVCRDRQLRKIKTNYFIV
SLAFADLLVSVLVMPFGAIELVQDIWVYGEMFCLVRTSLDVLLTTASIFHLCCISLDRYY
AICCQPLVYRNKMTPLRIALMLGGCWVIPMFISFLPIMQGWNNIGIVDLIEKRKFNQNSN
STYCVFMVNKPYAITCSVVAFYIPFLLMVLAYYRIYVTAKEHARQIQVLQRAGAPAEGRP
QPADQHSTHRMRTETKAAKTLCIIMGCFCLCWAPFFVTNIVDPFIDYTVPGQLWTAFLWL
GYINSGLNPFLYAFLNKSFRRAFLIILCCDDERYRRPSILGQTVPCSTTTINGSTHVLRD
TVECGGQWESQCHPAASSPLVAAQPIDT
|
|
|
|---|
| BDBM50005833 |
|---|
| n/a |
|---|
| Name | BDBM50005833 |
|---|
| Synonyms: | (exo)4-Amino-5-chloro-N-(hexahydro-pyrrolizin-1-ylmethyl)-2-methoxy-benzamide | 4-Amino-5-chloro-2-methoxy-N-(7a-methyl-hexahydro-pyrrolizin-1-ylmethyl)-benzamide | 4-Amino-5-chloro-N-(hexahydro-pyrrolizin-2-ylmethyl)-2-methoxy-benzamide | 4-Amino-5-chloro-N-[(1S,7aS)-1-(hexahydro-pyrrolizin-1-yl)methyl]-2-methoxy-benzamide | 4-amino-5-chloro-N-[(1S-cis-hexahydro-1H-pyrrolizin-1-yl)-methyl]-2-methoxybenzenecarboxamide | CHEMBL34291 | SC-53116 | SC49518 | SC53116 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H22ClN3O2 |
|---|
| Mol. Mass. | 323.818 |
|---|
| SMILES | COc1cc(N)c(Cl)cc1C(=O)NC[C@@H]1CCN2CCC[C@@H]12 |
|---|
| Structure | 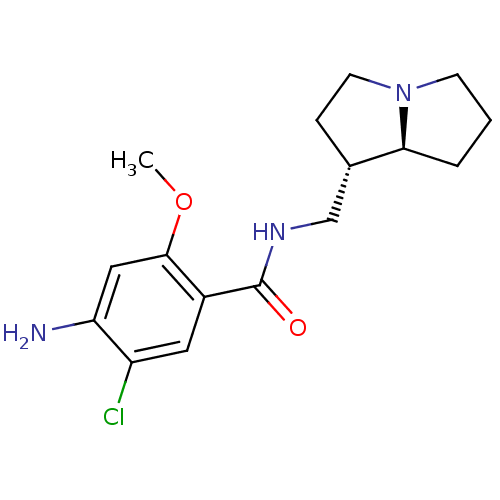
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Hirokawa, Y; Fujiwara, I; Suzuki, K; Harada, H; Yoshikawa, T; Yoshida, N; Kato, S Synthesis and structure-affinity relationships of novel N-(1-ethyl-4-methylhexahydro-1,4-diazepin-6-yl)pyridine-3-carboxamides with potent serotonin 5-HT3 and dopamine D2 receptor antagonistic activity. J Med Chem46:702-15 (2003) [PubMed] Article
Hirokawa, Y; Fujiwara, I; Suzuki, K; Harada, H; Yoshikawa, T; Yoshida, N; Kato, S Synthesis and structure-affinity relationships of novel N-(1-ethyl-4-methylhexahydro-1,4-diazepin-6-yl)pyridine-3-carboxamides with potent serotonin 5-HT3 and dopamine D2 receptor antagonistic activity. J Med Chem46:702-15 (2003) [PubMed] Article