| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen phosphorylase, muscle form |
|---|
| Ligand | BDBM50149315 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_138362 (CHEMBL749049) |
|---|
| Ki | 3000±n/a nM |
|---|
| Citation |  Kristiansen, M; Andersen, B; Iversen, LF; Westergaard, N Identification, synthesis, and characterization of new glycogen phosphorylase inhibitors binding to the allosteric AMP site. J Med Chem47:3537-45 (2004) [PubMed] Article Kristiansen, M; Andersen, B; Iversen, LF; Westergaard, N Identification, synthesis, and characterization of new glycogen phosphorylase inhibitors binding to the allosteric AMP site. J Med Chem47:3537-45 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen phosphorylase, muscle form |
|---|
| Name: | Glycogen phosphorylase, muscle form |
|---|
| Synonyms: | Glycogen phosphorylase, muscle form | Muscle glycogen phosphorylase | Myophosphorylase | PYGM | PYGM_HUMAN |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 97097.45 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1449751 |
|---|
| Residue: | 842 |
|---|
| Sequence: | MSRPLSDQEKRKQISVRGLAGVENVTELKKNFNRHLHFTLVKDRNVATPRDYYFALAHTV
RDHLVGRWIRTQQHYYEKDPKRIYYLSLEFYMGRTLQNTMVNLALENACDEATYQLGLDM
EELEEIEEDAGLGNGGLGRLAACFLDSMATLGLAAYGYGIRYEFGIFNQKISGGWQMEEA
DDWLRYGNPWEKARPEFTLPVHFYGHVEHTSQGAKWVDTQVVLAMPYDTPVPGYRNNVVN
TMRLWSAKAPNDFNLKDFNVGGYIQAVLDRNLAENISRVLYPNDNFFEGKELRLKQEYFV
VAATLQDIIRRFKSSKFGCRDPVRTNFDAFPDKVAIQLNDTHPSLAIPELMRILVDLERM
DWDKAWDVTVRTCAYTNHTVLPEALERWPVHLLETLLPRHLQIIYEINQRFLNRVAAAFP
GDVDRLRRMSLVEEGAVKRINMAHLCIAGSHAVNGVARIHSEILKKTIFKDFYELEPHKF
QNKTNGITPRRWLVLCNPGLAEVIAERIGEDFISDLDQLRKLLSFVDDEAFIRDVAKVKQ
ENKLKFAAYLEREYKVHINPNSLFDIQVKRIHEYKRQLLNCLHVITLYNRIKREPNKFFV
PRTVMIGGKAAPGYHMAKMIIRLVTAIGDVVNHDPAVGDRLRVIFLENYRVSLAEKVIPA
ADLSEQISTAGTEASGTGNMKFMLNGALTIGTMDGANVEMAEEAGEENFFIFGMRVEDVD
KLDQRGYNAQEYYDRIPELRQVIEQLSSGFFSPKQPDLFKDIVNMLMHHDRFKVFADYED
YIKCQEKVSALYKNPREWTRMVIRNIATSGKFSSDRTIAQYAREIWGVEPSRQRLPAPDE
AI
|
|
|
|---|
| BDBM50149315 |
|---|
| n/a |
|---|
| Name | BDBM50149315 |
|---|
| Synonyms: | (6S,7S,8R,9R)-6,7,8-Trihydroxy-9-hydroxymethyl-1,3-diaza-spiro[4.5]decane-2,4-dione | CHEMBL117003 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C9H14N2O6 |
|---|
| Mol. Mass. | 246.2173 |
|---|
| SMILES | OC[C@H]1C[C@@]2(NC(=O)NC2=O)[C@H](O)[C@@H](O)[C@@H]1O |
|---|
| Structure | 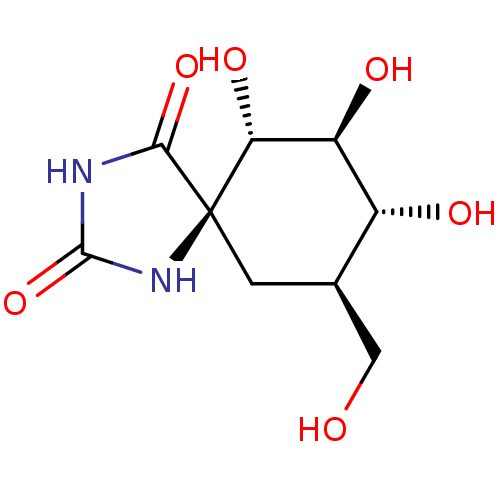
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kristiansen, M; Andersen, B; Iversen, LF; Westergaard, N Identification, synthesis, and characterization of new glycogen phosphorylase inhibitors binding to the allosteric AMP site. J Med Chem47:3537-45 (2004) [PubMed] Article
Kristiansen, M; Andersen, B; Iversen, LF; Westergaard, N Identification, synthesis, and characterization of new glycogen phosphorylase inhibitors binding to the allosteric AMP site. J Med Chem47:3537-45 (2004) [PubMed] Article