| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2a |
|---|
| Ligand | BDBM50229886 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_31369 (CHEMBL644661) |
|---|
| Ki | >1000±n/a nM |
|---|
| Citation |  Baraldi, PG; Tabrizi, MA; Preti, D; Bovero, A; Romagnoli, R; Fruttarolo, F; Zaid, NA; Moorman, AR; Varani, K; Gessi, S; Merighi, S; Borea, PA Design, synthesis, and biological evaluation of new 8-heterocyclic xanthine derivatives as highly potent and selective human A2B adenosine receptor antagonists. J Med Chem47:1434-47 (2004) [PubMed] Article Baraldi, PG; Tabrizi, MA; Preti, D; Bovero, A; Romagnoli, R; Fruttarolo, F; Zaid, NA; Moorman, AR; Varani, K; Gessi, S; Merighi, S; Borea, PA Design, synthesis, and biological evaluation of new 8-heterocyclic xanthine derivatives as highly potent and selective human A2B adenosine receptor antagonists. J Med Chem47:1434-47 (2004) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2a |
|---|
| Name: | Adenosine receptor A2a |
|---|
| Synonyms: | A2A adenosine receptor (hA2A) | AA2AR_HUMAN | ADENOSINE A2 | ADENOSINE A2a | ADORA2 | ADORA2A | Adenosine A2A receptor (A2AAR) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 44716.46 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P29274 |
|---|
| Residue: | 412 |
|---|
| Sequence: | MPIMGSSVYITVELAIAVLAILGNVLVCWAVWLNSNLQNVTNYFVVSLAAADIAVGVLAI
PFAITISTGFCAACHGCLFIACFVLVLTQSSIFSLLAIAIDRYIAIRIPLRYNGLVTGTR
AKGIIAICWVLSFAIGLTPMLGWNNCGQPKEGKNHSQGCGEGQVACLFEDVVPMNYMVYF
NFFACVLVPLLLMLGVYLRIFLAARRQLKQMESQPLPGERARSTLQKEVHAAKSLAIIVG
LFALCWLPLHIINCFTFFCPDCSHAPLWLMYLAIVLSHTNSVVNPFIYAYRIREFRQTFR
KIIRSHVLRQQEPFKAAGTSARVLAAHGSDGEQVSLRLNGHPPGVWANGSAPHPERRPNG
YALGLVSGGSAQESQGNTGLPDVELLSHELKGVCPEPPGLDDPLAQDGAGVS
|
|
|
|---|
| BDBM50229886 |
|---|
| n/a |
|---|
| Name | BDBM50229886 |
|---|
| Synonyms: | 2-(3,4-Dimethoxy-phenyl)-N-[5-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H-purin-8-yl)-1-methyl-1H-pyrazol-3-yl]-acetamide | 2-(3,4-dimethoxyphenyl)-N-(5-(2,6-dioxo-1,3-dipropyl-2,3,6,7-tetrahydro-1H-purin-8-yl)-1-methyl-1H-pyrazol-3-yl)acetamide | CHEMBL408186 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C25H31N7O5 |
|---|
| Mol. Mass. | 509.5575 |
|---|
| SMILES | CCCn1c2nc([nH]c2c(=O)n(CCC)c1=O)-c1cc(NC(=O)Cc2ccc(OC)c(OC)c2)nn1C |
|---|
| Structure | 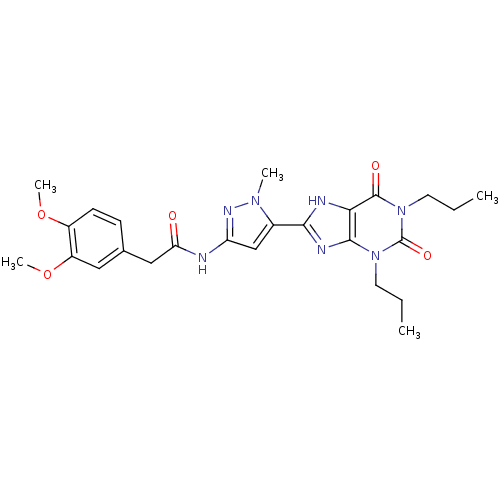
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Baraldi, PG; Tabrizi, MA; Preti, D; Bovero, A; Romagnoli, R; Fruttarolo, F; Zaid, NA; Moorman, AR; Varani, K; Gessi, S; Merighi, S; Borea, PA Design, synthesis, and biological evaluation of new 8-heterocyclic xanthine derivatives as highly potent and selective human A2B adenosine receptor antagonists. J Med Chem47:1434-47 (2004) [PubMed] Article
Baraldi, PG; Tabrizi, MA; Preti, D; Bovero, A; Romagnoli, R; Fruttarolo, F; Zaid, NA; Moorman, AR; Varani, K; Gessi, S; Merighi, S; Borea, PA Design, synthesis, and biological evaluation of new 8-heterocyclic xanthine derivatives as highly potent and selective human A2B adenosine receptor antagonists. J Med Chem47:1434-47 (2004) [PubMed] Article