| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mitogen-activated protein kinase 10 |
|---|
| Ligand | BDBM50169974 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_304976 (CHEMBL829309) |
|---|
| IC50 | 9400±n/a nM |
|---|
| Citation |  Gaillard, P; Jeanclaude-Etter, I; Ardissone, V; Arkinstall, S; Cambet, Y; Camps, M; Chabert, C; Church, D; Cirillo, R; Gretener, D; Halazy, S; Nichols, A; Szyndralewiez, C; Vitte, PA; Gotteland, JP Design and synthesis of the first generation of novel potent, selective, and in vivo active (benzothiazol-2-yl)acetonitrile inhibitors of the c-Jun N-terminal kinase. J Med Chem48:4596-607 (2005) [PubMed] Article Gaillard, P; Jeanclaude-Etter, I; Ardissone, V; Arkinstall, S; Cambet, Y; Camps, M; Chabert, C; Church, D; Cirillo, R; Gretener, D; Halazy, S; Nichols, A; Szyndralewiez, C; Vitte, PA; Gotteland, JP Design and synthesis of the first generation of novel potent, selective, and in vivo active (benzothiazol-2-yl)acetonitrile inhibitors of the c-Jun N-terminal kinase. J Med Chem48:4596-607 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mitogen-activated protein kinase 10 |
|---|
| Name: | Mitogen-activated protein kinase 10 |
|---|
| Synonyms: | Jnk3 | MK10_RAT | Mapk10 | Prkm10 | c-Jun N-terminal kinase 3 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 52534.34 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_1460661 |
|---|
| Residue: | 464 |
|---|
| Sequence: | MSLHFLYYCSEPTLDVKIAFCQGFDKHVDVSSVVKHYNMSKSKVDNQFYSVEVGDSTFTV
LKRYQNLKPIGSGAQGIVCAAYDAVLDRNVAIKKLSRPFQNQTHAKRAYRELVLMKCVNH
KNIISLLNVFTPQKTLEEFQDVYLVMELMDANLCQVIQMELDHERMSYLLYQMLCGIKHL
HSAGIIHRDLKPSNIVVKSDCTLKILDFGLARTAGTSFMMTPYVVTRYYRAPEVILGMGY
KENVDIWSVGCIMGEMVRHKILFPGRDYIDQWNKVIEQLGTPCPEFMKKLQPTVRNYVEN
RPKYAGLTFPKLFPDSLFPADSEHNKLKASQARDLLSKMLVIDPAKRISVDDALQHPYIN
VWYDPAEVEAPPPQIYDKQLDEREHTIEEWKELIYKEVMNSEEKTKNGVVKGQPSPSGAA
VNSSESLPPSSSVNDISSMSTDQTLASDTDSSLEASAGPLGCCR
|
|
|
|---|
| BDBM50169974 |
|---|
| n/a |
|---|
| Name | BDBM50169974 |
|---|
| Synonyms: | (2Z)-1,3-benzothiazol-2(3H)-ylidene[2-(dimethylamino)pyrimidin-4-yl]acetonitrile | CHEMBL190380 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C15H13N5S |
|---|
| Mol. Mass. | 295.362 |
|---|
| SMILES | CN(C)c1nccc(n1)C(C#N)c1nc2ccccc2s1 |
|---|
| Structure | 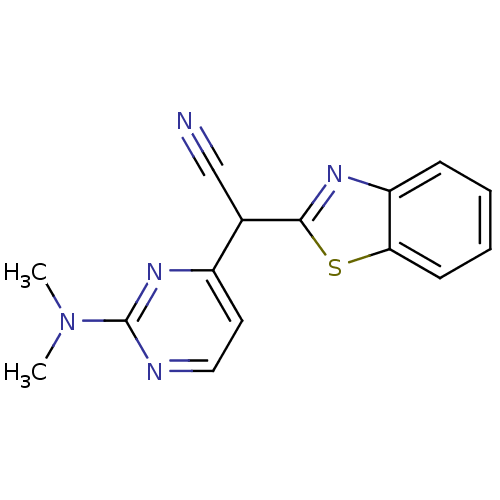
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gaillard, P; Jeanclaude-Etter, I; Ardissone, V; Arkinstall, S; Cambet, Y; Camps, M; Chabert, C; Church, D; Cirillo, R; Gretener, D; Halazy, S; Nichols, A; Szyndralewiez, C; Vitte, PA; Gotteland, JP Design and synthesis of the first generation of novel potent, selective, and in vivo active (benzothiazol-2-yl)acetonitrile inhibitors of the c-Jun N-terminal kinase. J Med Chem48:4596-607 (2005) [PubMed] Article
Gaillard, P; Jeanclaude-Etter, I; Ardissone, V; Arkinstall, S; Cambet, Y; Camps, M; Chabert, C; Church, D; Cirillo, R; Gretener, D; Halazy, S; Nichols, A; Szyndralewiez, C; Vitte, PA; Gotteland, JP Design and synthesis of the first generation of novel potent, selective, and in vivo active (benzothiazol-2-yl)acetonitrile inhibitors of the c-Jun N-terminal kinase. J Med Chem48:4596-607 (2005) [PubMed] Article