| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 6 |
|---|
| Ligand | BDBM50159204 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_303674 (CHEMBL830434) |
|---|
| Ki | 2±n/a nM |
|---|
| Citation |  Cole, DC; Lennox, WJ; Lombardi, S; Ellingboe, JW; Bernotas, RC; Tawa, GJ; Mazandarani, H; Smith, DL; Zhang, G; Coupet, J; Schechter, LE Discovery of 5-arylsulfonamido-3-(pyrrolidin-2-ylmethyl)-1H-indole derivatives as potent, selective 5-HT6 receptor agonists and antagonists. J Med Chem48:353-6 (2005) [PubMed] Article Cole, DC; Lennox, WJ; Lombardi, S; Ellingboe, JW; Bernotas, RC; Tawa, GJ; Mazandarani, H; Smith, DL; Zhang, G; Coupet, J; Schechter, LE Discovery of 5-arylsulfonamido-3-(pyrrolidin-2-ylmethyl)-1H-indole derivatives as potent, selective 5-HT6 receptor agonists and antagonists. J Med Chem48:353-6 (2005) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 6 |
|---|
| Name: | 5-hydroxytryptamine receptor 6 |
|---|
| Synonyms: | 5-HT-6 | 5-HT6 | 5-hydroxytryptamine receptor 6 (5-HT-6) | 5-hydroxytryptamine receptor 6 (5-HT6R) | 5-hydroxytryptamine receptor 6 (5HT6) | 5HT6R_HUMAN | HTR6 | Serotonin (5-HT3) receptor | Serotonin 6 (5-HT6) receptor | Serotonin Receptor 6 |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 46968.43 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50406 |
|---|
| Residue: | 440 |
|---|
| Sequence: | MVPEPGPTANSTPAWGAGPPSAPGGSGWVAAALCVVIALTAAANSLLIALICTQPALRNT
SNFFLVSLFTSDLMVGLVVMPPAMLNALYGRWVLARGLCLLWTAFDVMCCSASILNLCLI
SLDRYLLILSPLRYKLRMTPLRALALVLGAWSLAALASFLPLLLGWHELGHARPPVPGQC
RLLASLPFVLVASGLTFFLPSGAICFTYCRILLAARKQAVQVASLTTGMASQASETLQVP
RTPRPGVESADSRRLATKHSRKALKASLTLGILLGMFFVTWLPFFVANIVQAVCDCISPG
LFDVLTWLGYCNSTMNPIIYPLFMRDFKRALGRFLPCPRCPRERQASLASPSLRTSHSGP
RPGLSLQQVLPLPLPPDSDSDSDAGSGGSSGLRLTAQLLLPGEATQDPPLPTRAAAAVNF
FNIDPAEPELRPHPLGIPTN
|
|
|
|---|
| BDBM50159204 |
|---|
| n/a |
|---|
| Name | BDBM50159204 |
|---|
| Synonyms: | 5-Chloro-3-methyl-benzo[b]thiophene-2-sulfonic acid [3-((S)-1-methyl-pyrrolidin-2-ylmethyl)-1H-indol-5-yl]-amide |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C23H24ClN3O2S2 |
|---|
| Mol. Mass. | 474.039 |
|---|
| SMILES | CN1CCCC1Cc1c[nH]c2ccc(NS(=O)(=O)c3sc4ccc(Cl)cc4c3C)cc12 |
|---|
| Structure | 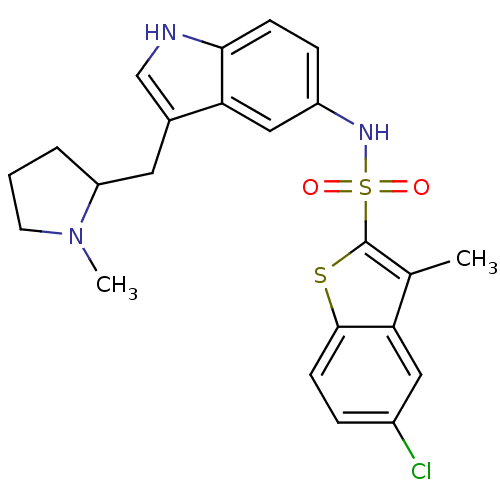
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Cole, DC; Lennox, WJ; Lombardi, S; Ellingboe, JW; Bernotas, RC; Tawa, GJ; Mazandarani, H; Smith, DL; Zhang, G; Coupet, J; Schechter, LE Discovery of 5-arylsulfonamido-3-(pyrrolidin-2-ylmethyl)-1H-indole derivatives as potent, selective 5-HT6 receptor agonists and antagonists. J Med Chem48:353-6 (2005) [PubMed] Article
Cole, DC; Lennox, WJ; Lombardi, S; Ellingboe, JW; Bernotas, RC; Tawa, GJ; Mazandarani, H; Smith, DL; Zhang, G; Coupet, J; Schechter, LE Discovery of 5-arylsulfonamido-3-(pyrrolidin-2-ylmethyl)-1H-indole derivatives as potent, selective 5-HT6 receptor agonists and antagonists. J Med Chem48:353-6 (2005) [PubMed] Article