| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Mu-type opioid receptor |
|---|
| Ligand | BDBM50019056 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_378702 (CHEMBL853450) |
|---|
| Ki | 6.9±n/a nM |
|---|
| Citation |  Crooks, PA; Kottayil, SG; Al-Ghananeem, AM; Byrn, SR; Butterfield, DA Opiate receptor binding properties of morphine-, dihydromorphine-, and codeine 6-O-sulfate ester congeners. Bioorg Med Chem Lett16:4291-5 (2006) [PubMed] Article Crooks, PA; Kottayil, SG; Al-Ghananeem, AM; Byrn, SR; Butterfield, DA Opiate receptor binding properties of morphine-, dihydromorphine-, and codeine 6-O-sulfate ester congeners. Bioorg Med Chem Lett16:4291-5 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Mu-type opioid receptor |
|---|
| Name: | Mu-type opioid receptor |
|---|
| Synonyms: | M-OR-1 | MOR-1 | Mu opioid receptor | Mu-type opioid receptor (Mu) | OPIATE Mu | OPRM1 | OPRM_CAVPO |
|---|
| Type: | Enzyme Catalytic Domain |
|---|
| Mol. Mass.: | 11165.58 |
|---|
| Organism: | GUINEA PIG |
|---|
| Description: | P97266 |
|---|
| Residue: | 98 |
|---|
| Sequence: | YTKMKTATNIYIFNLALADALATSTLPFQSVNYLMGTWPFGTILCKIVISIDYYNMFTSI
FTLCTMSVDRYIAVCHPVKALDFRTPRNAKTVNVCNWI
|
|
|
|---|
| BDBM50019056 |
|---|
| n/a |
|---|
| Name | BDBM50019056 |
|---|
| Synonyms: | (S)-2-[(S)-2-(2-{2-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-acetylamino}-acetylamino)-3-phenyl-propionylamino]-4-methylsulfanyl-butyric acid | ([Met]-enkephalin)2-[2-(2-{2-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-acetylamino}-acetylamino)-3-phenyl-propionylamino]-4-methylsulfanyl-butyric acid([Met]-enkephalin | 2-[(S)-2-(2-{2-[(S)-2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-acetylamino}-acetylamino)-1-oxo-3-phenyl-propylamino]-4-methylsulfanyl-butyric acid | 2-[1-[1-amino-2-(4-hydroxyphenyl)-(1S)-ethylcarboxamidomethylcarboxamidomethylcarboxamido]-2-phenyl-(1S)-ethylcarboxamido]-4-methylsulfanyl-(2S)-butanoic acid | 2-[2-(2-{2-[2-Amino-3-(4-hydroxy-phenyl)-propionylamino]-acetylamino}-acetylamino)-3-phenyl-propionylamino]-4-methylsulfanyl-butyric acid | CHEMBL13786 | H-Tyr-Gly-Gly-Phe-Met-OH | Met5-Enkephalin | [Met5]-enkephalin | met-enkephalin |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C27H35N5O7S |
|---|
| Mol. Mass. | 573.661 |
|---|
| SMILES | CSCC[C@H](NC(=O)[C@H](Cc1ccccc1)NC(=O)CNC(=O)CNC(=O)[C@@H](N)Cc1ccc(O)cc1)C(O)=O |
|---|
| Structure | 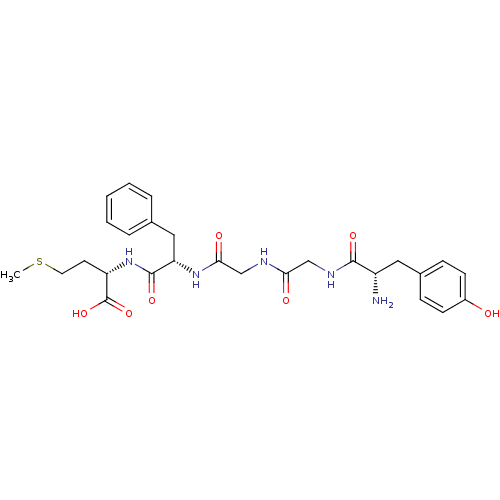
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Crooks, PA; Kottayil, SG; Al-Ghananeem, AM; Byrn, SR; Butterfield, DA Opiate receptor binding properties of morphine-, dihydromorphine-, and codeine 6-O-sulfate ester congeners. Bioorg Med Chem Lett16:4291-5 (2006) [PubMed] Article
Crooks, PA; Kottayil, SG; Al-Ghananeem, AM; Byrn, SR; Butterfield, DA Opiate receptor binding properties of morphine-, dihydromorphine-, and codeine 6-O-sulfate ester congeners. Bioorg Med Chem Lett16:4291-5 (2006) [PubMed] Article