| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Adenosine receptor A2b |
|---|
| Ligand | BDBM50202573 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_423827 (CHEMBL911272) |
|---|
| EC50 | >5000±n/a nM |
|---|
| Citation |  Baraldi, PG; Preti, D; Tabrizi, MA; Fruttarolo, F; Romagnoli, R; Carrion, MD; Cara, LC; Moorman, AR; Varani, K; Borea, PA Synthesis and biological evaluation of novel 1-deoxy-1-[6-[((hetero)arylcarbonyl)hydrazino]- 9H-purin-9-yl]-N-ethyl-beta-D-ribofuranuronamide derivatives as useful templates for the development of A2B adenosine receptor agonists. J Med Chem50:374-80 (2007) [PubMed] Article Baraldi, PG; Preti, D; Tabrizi, MA; Fruttarolo, F; Romagnoli, R; Carrion, MD; Cara, LC; Moorman, AR; Varani, K; Borea, PA Synthesis and biological evaluation of novel 1-deoxy-1-[6-[((hetero)arylcarbonyl)hydrazino]- 9H-purin-9-yl]-N-ethyl-beta-D-ribofuranuronamide derivatives as useful templates for the development of A2B adenosine receptor agonists. J Med Chem50:374-80 (2007) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Adenosine receptor A2b |
|---|
| Name: | Adenosine receptor A2b |
|---|
| Synonyms: | AA2BR_HUMAN | ADENOSINE A2B | ADORA2B | Adenosine receptor A2B (A2B) | Adenosine receptors A2b | Adenosine receptors; A2a & A2b |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 36341.22 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 332 |
|---|
| Sequence: | MLLETQDALYVALELVIAALSVAGNVLVCAAVGTANTLQTPTNYFLVSLAAADVAVGLFA
IPFAITISLGFCTDFYGCLFLACFVLVLTQSSIFSLLAVAVDRYLAICVPLRYKSLVTGT
RARGVIAVLWVLAFGIGLTPFLGWNSKDSATNNCTEPWDGTTNESCCLVKCLFENVVPMS
YMVYFNFFGCVLPPLLIMLVIYIKIFLVACRQLQRTELMDHSRTTLQREIHAAKSLAMIV
GIFALCWLPVHAVNCVTLFQPAQGKNKPKWAMNMAILLSHANSVVNPIVYAYRNRDFRYT
FHKIISRYLLCQADVKSGNGQAGVQPALGVGL
|
|
|
|---|
| BDBM50202573 |
|---|
| n/a |
|---|
| Name | BDBM50202573 |
|---|
| Synonyms: | 1-deoxy-1-{6-[N'-(pyridine-3-carbonyl)-hydrazino]-9H-purin-9-yl}-N-ethyl-beta-D-ribofuranuronamide | CHEMBL448482 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H20N8O5 |
|---|
| Mol. Mass. | 428.402 |
|---|
| SMILES | CCNC(=O)[C@H]1O[C@H]([C@H](O)[C@@H]1O)n1cnc2c(NNC(=O)c3cccnc3)ncnc12 |r| |
|---|
| Structure | 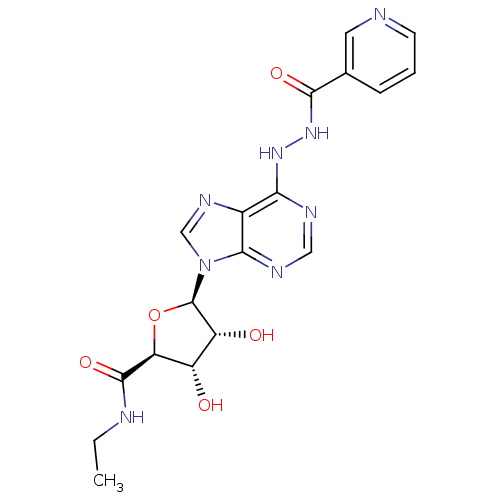
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Baraldi, PG; Preti, D; Tabrizi, MA; Fruttarolo, F; Romagnoli, R; Carrion, MD; Cara, LC; Moorman, AR; Varani, K; Borea, PA Synthesis and biological evaluation of novel 1-deoxy-1-[6-[((hetero)arylcarbonyl)hydrazino]- 9H-purin-9-yl]-N-ethyl-beta-D-ribofuranuronamide derivatives as useful templates for the development of A2B adenosine receptor agonists. J Med Chem50:374-80 (2007) [PubMed] Article
Baraldi, PG; Preti, D; Tabrizi, MA; Fruttarolo, F; Romagnoli, R; Carrion, MD; Cara, LC; Moorman, AR; Varani, K; Borea, PA Synthesis and biological evaluation of novel 1-deoxy-1-[6-[((hetero)arylcarbonyl)hydrazino]- 9H-purin-9-yl]-N-ethyl-beta-D-ribofuranuronamide derivatives as useful templates for the development of A2B adenosine receptor agonists. J Med Chem50:374-80 (2007) [PubMed] Article