| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Peroxisome proliferator-activated receptor alpha |
|---|
| Ligand | BDBM50371235 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_459972 (CHEMBL943100) |
|---|
| EC50 | >10000±n/a nM |
|---|
| Citation |  Matthews, JM; Chen, X; Cryan, E; Hlasta, DJ; Rybczynski, PJ; Strauss, K; Tang, Y; Xu, JZ; Yang, M; Zhou, L; Demarest, KT Design and synthesis of indane-ureido-thioisobutyric acids: A novel class of PPARalpha agonists. Bioorg Med Chem Lett17:6773-8 (2008) [PubMed] Article Matthews, JM; Chen, X; Cryan, E; Hlasta, DJ; Rybczynski, PJ; Strauss, K; Tang, Y; Xu, JZ; Yang, M; Zhou, L; Demarest, KT Design and synthesis of indane-ureido-thioisobutyric acids: A novel class of PPARalpha agonists. Bioorg Med Chem Lett17:6773-8 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Peroxisome proliferator-activated receptor alpha |
|---|
| Name: | Peroxisome proliferator-activated receptor alpha |
|---|
| Synonyms: | Nr1c1 | Nuclear receptor subfamily 1 group C member 1 | PPAR-alpha | PPARA_RAT | Peroxisome Proliferator-Activated Receptor alpha | Ppar | Ppara |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 52374.28 |
|---|
| Organism: | Rattus norvegicus |
|---|
| Description: | ChEMBL_834194 |
|---|
| Residue: | 468 |
|---|
| Sequence: | MVDTESPICPLSPLEADDLESPLSEEFLQEMGNIQEISQSLGEESSGSFSFADYQYLGSC
PGSEGSVITDTLSPASSPSSVSCPAVPTSTDESPGNALNIECRICGDKASGYHYGVHACE
GCKGFFRRTIRLKLAYDKCDRSCKIQKKNRNKCQYCRFHKCLSVGMSHNAIRFGRMPRSE
KAKLKAEILTCEHDLKDSETADLKSLAKRIHEAYLKNFNMNKVKARVILAGKTSNNPPFV
IHDMETLCMAEKTLVAKMVANGVENKEAEVRFFHCCQCMSVETVTELTEFAKAIPGFANL
DLNDQVTLLKYGVYEAIFTMLSSLMNKDGMLIAYGNGFITREFLKNLRKPFCDIMEPKFD
FAMKFNALELDDSDISLFVAAIICCGDRPGLLNIGYIEKLQEGIVHVLKLHLQSNHPDDT
FLFPKLLQKMVDLRQLVTEHAQLVQVIKKTESDAALHPLLQEIYRDMY
|
|
|
|---|
| BDBM50371235 |
|---|
| n/a |
|---|
| Name | BDBM50371235 |
|---|
| Synonyms: | CIPROFIBRATE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C13H14Cl2O3 |
|---|
| Mol. Mass. | 289.154 |
|---|
| SMILES | CC(C)(Oc1ccc(cc1)C1CC1(Cl)Cl)C(O)=O |
|---|
| Structure | 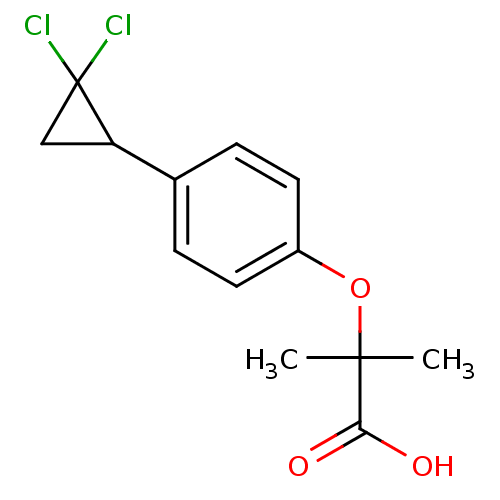
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Matthews, JM; Chen, X; Cryan, E; Hlasta, DJ; Rybczynski, PJ; Strauss, K; Tang, Y; Xu, JZ; Yang, M; Zhou, L; Demarest, KT Design and synthesis of indane-ureido-thioisobutyric acids: A novel class of PPARalpha agonists. Bioorg Med Chem Lett17:6773-8 (2008) [PubMed] Article
Matthews, JM; Chen, X; Cryan, E; Hlasta, DJ; Rybczynski, PJ; Strauss, K; Tang, Y; Xu, JZ; Yang, M; Zhou, L; Demarest, KT Design and synthesis of indane-ureido-thioisobutyric acids: A novel class of PPARalpha agonists. Bioorg Med Chem Lett17:6773-8 (2008) [PubMed] Article