| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Prolyl endopeptidase |
|---|
| Ligand | BDBM50374386 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_469247 (CHEMBL948947) |
|---|
| IC50 | 5.6±n/a nM |
|---|
| Citation |  Maryanoff, BE; Costanzo, MJ Inhibitors of proteases and amide hydrolases that employ an alpha-ketoheterocycle as a key enabling functionality. Bioorg Med Chem16:1562-95 (2008) [PubMed] Article Maryanoff, BE; Costanzo, MJ Inhibitors of proteases and amide hydrolases that employ an alpha-ketoheterocycle as a key enabling functionality. Bioorg Med Chem16:1562-95 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Prolyl endopeptidase |
|---|
| Name: | Prolyl endopeptidase |
|---|
| Synonyms: | 3.4.21.26 | PE | PPCE_PIG | PREP | Post-proline cleaving enzyme |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 80758.04 |
|---|
| Organism: | Sus scrofa |
|---|
| Description: | n/a |
|---|
| Residue: | 710 |
|---|
| Sequence: | MLSFQYPDVYRDETAIQDYHGHKVCDPYAWLEDPDSEQTKAFVEAQNKITVPFLEQCPIR
GLYKERMTELYDYPKYSCHFKKGKRYFYFYNTGLQNQRVLYVQDSLEGEARVFLDPNILS
DDGTVALRGYAFSEDGEYFAYGLSASGSDWVTIKFMKVDGAKELPDVLERVKFSCMAWTH
DGKGMFYNAYPQQDGKSDGTETSTNLHQKLYYHVLGTDQSEDILCAEFPDEPKWMGGAEL
SDDGRYVLLSIREGCDPVNRLWYCDLQQESNGITGILKWVKLIDNFEGEYDYVTNEGTVF
TFKTNRHSPNYRLINIDFTDPEESKWKVLVPEHEKDVLEWVACVRSNFLVLCYLHDVKNT
LQLHDLATGALLKIFPLEVGSVVGYSGQKKDTEIFYQFTSFLSPGIIYHCDLTKEELEPR
VFREVTVKGIDASDYQTVQIFYPSKDGTKIPMFIVHKKGIKLDGSHPAFLYGYGGFNISI
TPNYSVSRLIFVRHMGGVLAVANIRGGGEYGETWHKGGILANKQNCFDDFQCAAEYLIKE
GYTSPKRLTINGGSNGGLLVATCANQRPDLFGCVIAQVGVMDMLKFHKYTIGHAWTTDYG
CSDSKQHFEWLIKYSPLHNVKLPEADDIQYPSMLLLTADHDDRVVPLHSLKFIATLQYIV
GRSRKQNNPLLIHVDTKAGHGAGKPTAKVIEEVSDMFAFIARCLNIDWIP
|
|
|
|---|
| BDBM50374386 |
|---|
| n/a |
|---|
| Name | BDBM50374386 |
|---|
| Synonyms: | CHEMBL269970 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C26H29N3O3 |
|---|
| Mol. Mass. | 431.5268 |
|---|
| SMILES | O=C([C@@H]1CCCN1CCCc1ccccc1)N1CCC[C@H]1C(=O)c1nc2ccccc2o1 |
|---|
| Structure | 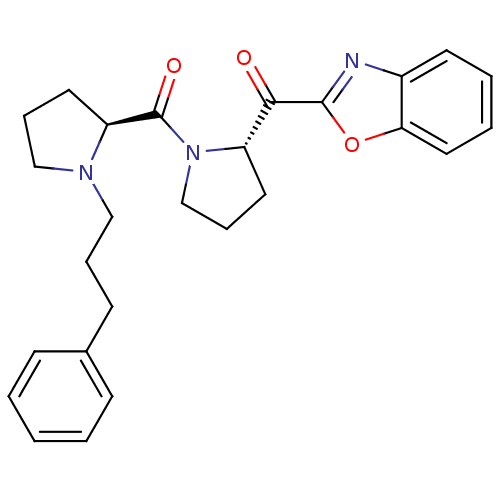
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Maryanoff, BE; Costanzo, MJ Inhibitors of proteases and amide hydrolases that employ an alpha-ketoheterocycle as a key enabling functionality. Bioorg Med Chem16:1562-95 (2008) [PubMed] Article
Maryanoff, BE; Costanzo, MJ Inhibitors of proteases and amide hydrolases that employ an alpha-ketoheterocycle as a key enabling functionality. Bioorg Med Chem16:1562-95 (2008) [PubMed] Article