| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A4 |
|---|
| Ligand | BDBM50259677 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_528535 (CHEMBL972070) |
|---|
| IC50 | 13400±n/a nM |
|---|
| Citation |  Gafner, S; Dietz, BM; McPhail, KL; Scott, IM; Glinski, JA; Russell, FE; McCollom, MM; Budzinski, JW; Foster, BC; Bergeron, C; Rhyu, MR; Bolton, JL Alkaloids from Eschscholzia californica and their capacity to inhibit binding of [3H]8-Hydroxy-2-(di-N-propylamino)tetralin to 5-HT1A receptors in Vitro. J Nat Prod69:432-5 (2006) [PubMed] Article Gafner, S; Dietz, BM; McPhail, KL; Scott, IM; Glinski, JA; Russell, FE; McCollom, MM; Budzinski, JW; Foster, BC; Bergeron, C; Rhyu, MR; Bolton, JL Alkaloids from Eschscholzia californica and their capacity to inhibit binding of [3H]8-Hydroxy-2-(di-N-propylamino)tetralin to 5-HT1A receptors in Vitro. J Nat Prod69:432-5 (2006) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A4 |
|---|
| Name: | Cytochrome P450 3A4 |
|---|
| Synonyms: | Albendazole monooxygenase | Albendazole sulfoxidase | CP3A4_HUMAN | CYP3A3 | CYP3A4 | CYPIIIA3 | CYPIIIA4 | Cytochrome P450 3A3 | Cytochrome P450 3A4 (CYP3A4) | Cytochrome P450 HLp | Nifedipine oxidase | Quinine 3-monooxygenase | Taurochenodeoxycholate 6-alpha-hydroxylase |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 57349.57 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 503 |
|---|
| Sequence: | MALIPDLAMETWLLLAVSLVLLYLYGTHSHGLFKKLGIPGPTPLPFLGNILSYHKGFCMF
DMECHKKYGKVWGFYDGQQPVLAITDPDMIKTVLVKECYSVFTNRRPFGPVGFMKSAISI
AEDEEWKRLRSLLSPTFTSGKLKEMVPIIAQYGDVLVRNLRREAETGKPVTLKDVFGAYS
MDVITSTSFGVNIDSLNNPQDPFVENTKKLLRFDFLDPFFLSITVFPFLIPILEVLNICV
FPREVTNFLRKSVKRMKESRLEDTQKHRVDFLQLMIDSQNSKETESHKALSDLELVAQSI
IFIFAGYETTSSVLSFIMYELATHPDVQQKLQEEIDAVLPNKAPPTYDTVLQMEYLDMVV
NETLRLFPIAMRLERVCKKDVEINGMFIPKGVVVMIPSYALHRDPKYWTEPEKFLPERFS
KKNKDNIDPYIYTPFGSGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLSLG
GLLQPEKPVVLKVESRDGTVSGA
|
|
|
|---|
| BDBM50259677 |
|---|
| n/a |
|---|
| Name | BDBM50259677 |
|---|
| Synonyms: | CHEMBL481839 | escholtzine |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H17NO4 |
|---|
| Mol. Mass. | 323.3426 |
|---|
| SMILES | CN1[C@H]2Cc3cc4OCOc4cc3[C@@H]1Cc1cc3OCOc3cc21 |r,TLB:0:1:14.15.23:3.12.4| |
|---|
| Structure | 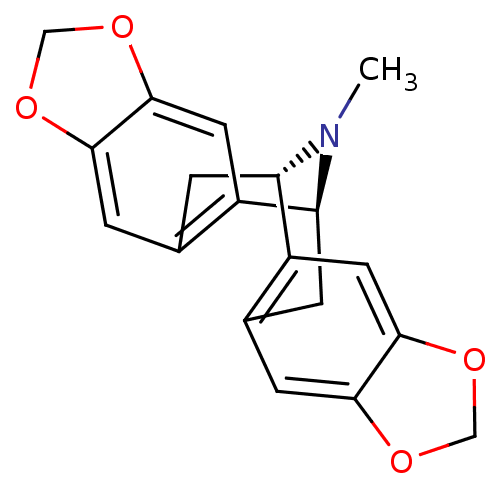
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Gafner, S; Dietz, BM; McPhail, KL; Scott, IM; Glinski, JA; Russell, FE; McCollom, MM; Budzinski, JW; Foster, BC; Bergeron, C; Rhyu, MR; Bolton, JL Alkaloids from Eschscholzia californica and their capacity to inhibit binding of [3H]8-Hydroxy-2-(di-N-propylamino)tetralin to 5-HT1A receptors in Vitro. J Nat Prod69:432-5 (2006) [PubMed] Article
Gafner, S; Dietz, BM; McPhail, KL; Scott, IM; Glinski, JA; Russell, FE; McCollom, MM; Budzinski, JW; Foster, BC; Bergeron, C; Rhyu, MR; Bolton, JL Alkaloids from Eschscholzia californica and their capacity to inhibit binding of [3H]8-Hydroxy-2-(di-N-propylamino)tetralin to 5-HT1A receptors in Vitro. J Nat Prod69:432-5 (2006) [PubMed] Article