| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Glycogen synthase kinase-3 beta |
|---|
| Ligand | BDBM50162083 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_501673 (CHEMBL981421) |
|---|
| Ki | 22±n/a nM |
|---|
| Citation |  Pollard, JR; Mortimore, M Discovery and development of aurora kinase inhibitors as anticancer agents. J Med Chem52:2629-51 (2009) [PubMed] Article Pollard, JR; Mortimore, M Discovery and development of aurora kinase inhibitors as anticancer agents. J Med Chem52:2629-51 (2009) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Glycogen synthase kinase-3 beta |
|---|
| Name: | Glycogen synthase kinase-3 beta |
|---|
| Synonyms: | GSK-3 beta | GSK-3, beta | GSK3B | GSK3B_HUMAN | Glycogen synthase kinase 3 beta (GSK3B) | Glycogen synthase kinase 3-beta (GSK3B) | Glycogen synthase kinase-3 beta (GSK-3B) | Glycogen synthase kinase-3 beta (GSK3 Beta) | Glycogen synthase kinase-3 beta (GSK3B) | Glycogen synthase kinase-3B (GSK-3B) | Glycogen synthase kinase-3beta (GSK3B) | Serine/threonine-protein kinase GSK3B |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 46756.38 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P49841 |
|---|
| Residue: | 420 |
|---|
| Sequence: | MSGRPRTTSFAESCKPVQQPSAFGSMKVSRDKDGSKVTTVVATPGQGPDRPQEVSYTDTK
VIGNGSFGVVYQAKLCDSGELVAIKKVLQDKRFKNRELQIMRKLDHCNIVRLRYFFYSSG
EKKDEVYLNLVLDYVPETVYRVARHYSRAKQTLPVIYVKLYMYQLFRSLAYIHSFGICHR
DIKPQNLLLDPDTAVLKLCDFGSAKQLVRGEPNVSYICSRYYRAPELIFGATDYTSSIDV
WSAGCVLAELLLGQPIFPGDSGVDQLVEIIKVLGTPTREQIREMNPNYTEFKFPQIKAHP
WTKVFRPRTPPEAIALCSRLLEYTPTARLTPLEACAHSFFDELRDPNVKLPNGRDTPALF
NFTTQELSSNPPLATILIPPHARIQAAASTPTNATAASDANTGDRGQTNNAASASASNST
|
|
|
|---|
| BDBM50162083 |
|---|
| n/a |
|---|
| Name | BDBM50162083 |
|---|
| Synonyms: | (5-Methyl-1H-pyrazol-3-yl)-(2-phenyl-quinazolin-4-yl)-amine | (5-Methyl-1H-pyrazol-3-yl)-(2-phenylquinazolin-4-yl)amine | CHEMBL359482 | N-(3-methyl-1H-pyrazol-5-yl)-2-phenylquinazolin-4-amine | N-(5-METHYL-1H-PYRAZOL-3-YL)-2-PHENYLQUINAZOLIN-4-AMINE |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H15N5 |
|---|
| Mol. Mass. | 301.3452 |
|---|
| SMILES | Cc1cc(Nc2nc(nc3ccccc23)-c2ccccc2)[nH]n1 |
|---|
| Structure | 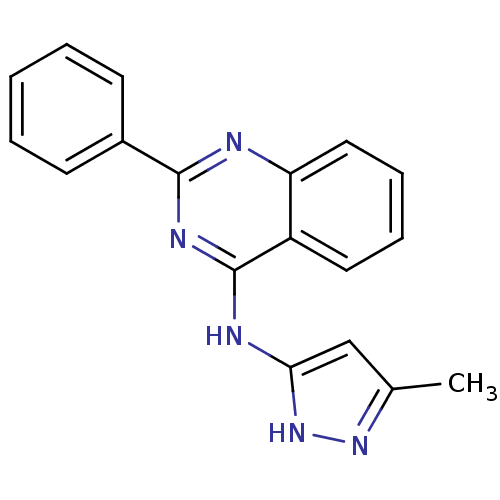
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Pollard, JR; Mortimore, M Discovery and development of aurora kinase inhibitors as anticancer agents. J Med Chem52:2629-51 (2009) [PubMed] Article
Pollard, JR; Mortimore, M Discovery and development of aurora kinase inhibitors as anticancer agents. J Med Chem52:2629-51 (2009) [PubMed] Article