| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 3A |
|---|
| Ligand | BDBM50307829 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_612615 (CHEMBL1065652) |
|---|
| Ki | 2.8±n/a nM |
|---|
| Citation |  Vernekar, SK; Hallaq, HY; Clarkson, G; Thompson, AJ; Silvestri, L; Lummis, SC; Lochner, M Toward biophysical probes for the 5-HT3 receptor: structure-activity relationship study of granisetron derivatives. J Med Chem53:2324-8 (2010) [PubMed] Article Vernekar, SK; Hallaq, HY; Clarkson, G; Thompson, AJ; Silvestri, L; Lummis, SC; Lochner, M Toward biophysical probes for the 5-HT3 receptor: structure-activity relationship study of granisetron derivatives. J Med Chem53:2324-8 (2010) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 3A |
|---|
| Name: | 5-hydroxytryptamine receptor 3A |
|---|
| Synonyms: | 5-HT-3 | 5-HT3 | 5-HT3A | 5-HT3R | 5-hydroxytryptamine receptor 3 (5-HT3) | 5-hydroxytryptamine receptor 3A (5-HT3a) | 5-hydroxytryptamine receptor 3A (5HT3A) | 5HT3A_HUMAN | 5HT3R | HTR3 | HTR3A | Serotonin 3 (5-HT3) receptor | Serotonin 3 receptor (5HT3) | Serotonin receptor 3A | Serotonin-gated ion channel receptor | Serotonin-gated ion channel receptor 3 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55283.27 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P46098 |
|---|
| Residue: | 478 |
|---|
| Sequence: | MLLWVQQALLALLLPTLLAQGEARRSRNTTRPALLRLSDYLLTNYRKGVRPVRDWRKPTT
VSIDVIVYAILNVDEKNQVLTTYIWYRQYWTDEFLQWNPEDFDNITKLSIPTDSIWVPDI
LINEFVDVGKSPNIPYVYIRHQGEVQNYKPLQVVTACSLDIYNFPFDVQNCSLTFTSWLH
TIQDINISLWRLPEKVKSDRSVFMNQGEWELLGVLPYFREFSMESSNYYAEMKFYVVIRR
RPLFYVVSLLLPSIFLMVMDIVGFYLPPNSGERVSFKITLLLGYSVFLIIVSDTLPATAI
GTPLIGVYFVVCMALLVISLAETIFIVRLVHKQDLQQPVPAWLRHLVLERIAWLLCLREQ
STSQRPPATSQATKTDDCSAMGNHCSHMGGPQDFEKSPRDRCSPPPPPREASLAVCGLLQ
ELSSIRQFLEKRDEIREVARDWLRVGSVLDKLLFHIYLLAVLAYSITLVMLWSIWQYA
|
|
|
|---|
| BDBM50307829 |
|---|
| n/a |
|---|
| Name | BDBM50307829 |
|---|
| Synonyms: | 5,5-difluoro-7,9-dimethyl-3-(2-(3-(3-((1S,5S)-9-methyl-9-azabicyclo[3.3.1]nonan-3-ylcarbamoyl)-1H-indazol-1-yl)propylamino)-2-oxoethylamino)-5H-dipyrrolo[1,2-c:1',2'-f][1,3,2]diazaborinin-4-ium-5-uide | CHEMBL592168 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C33H41BF2N8O2 |
|---|
| Mol. Mass. | 630.539 |
|---|
| SMILES | CN1C2CCCC1CC(C2)NC(=O)c1nn(CCCNC(=O)C\[NH+]=C2\C=CC3=Cc4c(C)cc(C)n4[B-](F)(F)N23)c2ccccc12 |c:27,t:29| |
|---|
| Structure | 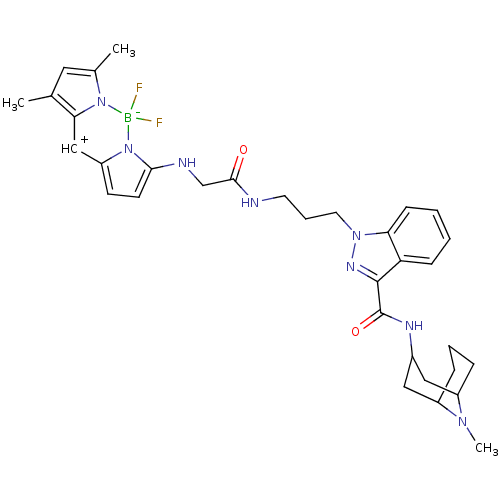
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Vernekar, SK; Hallaq, HY; Clarkson, G; Thompson, AJ; Silvestri, L; Lummis, SC; Lochner, M Toward biophysical probes for the 5-HT3 receptor: structure-activity relationship study of granisetron derivatives. J Med Chem53:2324-8 (2010) [PubMed] Article
Vernekar, SK; Hallaq, HY; Clarkson, G; Thompson, AJ; Silvestri, L; Lummis, SC; Lochner, M Toward biophysical probes for the 5-HT3 receptor: structure-activity relationship study of granisetron derivatives. J Med Chem53:2324-8 (2010) [PubMed] Article