| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 2E1 |
|---|
| Ligand | BDBM50396352 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_861871 (CHEMBL2173527) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Díaz, JL; Cuberes, R; Berrocal, J; Contijoch, M; Christmann, U; Fernández, A; Port, A; Holenz, J; Buschmann, H; Laggner, C; Serafini, MT; Burgueño, J; Zamanillo, D; Merlos, M; Vela, JM; Almansa, C Synthesis and biological evaluation of the 1-arylpyrazole class ofs(1) receptor antagonists: identification of 4-{2-[5-methyl-1-(naphthalen-2-yl)-1H-pyrazol-3-yloxy]ethyl}morpholine (S1RA, E-52862). J Med Chem55:8211-24 (2012) [PubMed] Article Díaz, JL; Cuberes, R; Berrocal, J; Contijoch, M; Christmann, U; Fernández, A; Port, A; Holenz, J; Buschmann, H; Laggner, C; Serafini, MT; Burgueño, J; Zamanillo, D; Merlos, M; Vela, JM; Almansa, C Synthesis and biological evaluation of the 1-arylpyrazole class ofs(1) receptor antagonists: identification of 4-{2-[5-methyl-1-(naphthalen-2-yl)-1H-pyrazol-3-yloxy]ethyl}morpholine (S1RA, E-52862). J Med Chem55:8211-24 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 2E1 |
|---|
| Name: | Cytochrome P450 2E1 |
|---|
| Synonyms: | CP2E1_HUMAN | CYP2E | CYP2E1 | Cytochrome P450 2E1 (CYP2E1) |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 56860.34 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P05181 |
|---|
| Residue: | 493 |
|---|
| Sequence: | MSALGVTVALLVWAAFLLLVSMWRQVHSSWNLPPGPFPLPIIGNLFQLELKNIPKSFTRL
AQRFGPVFTLYVGSQRMVVMHGYKAVKEALLDYKDEFSGRGDLPAFHAHRDRGIIFNNGP
TWKDIRRFSLTTLRNYGMGKQGNESRIQREAHFLLEALRKTQGQPFDPTFLIGCAPCNVI
ADILFRKHFDYNDEKFLRLMYLFNENFHLLSTPWLQLYNNFPSFLHYLPGSHRKVIKNVA
EVKEYVSERVKEHHQSLDPNCPRDLTDCLLVEMEKEKHSAERLYTMDGITVTVADLFFAG
TETTSTTLRYGLLILMKYPEIEEKLHEEIDRVIGPSRIPAIKDRQEMPYMDAVVHEIQRF
ITLVPSNLPHEATRDTIFRGYLIPKGTVVVPTLDSVLYDNQEFPDPEKFKPEHFLNENGK
FKYSDYFKPFSTGKRVCAGEGLARMELFLLLCAILQHFNLKPLVDPKDIDLSPIHIGFGC
IPPRYKLCVIPRS
|
|
|
|---|
| BDBM50396352 |
|---|
| n/a |
|---|
| Name | BDBM50396352 |
|---|
| Synonyms: | CHEMBL2170062 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C20H23N3O2 |
|---|
| Mol. Mass. | 337.4155 |
|---|
| SMILES | Cc1cc(OCCN2CCOCC2)nn1-c1ccc2ccccc2c1 |
|---|
| Structure | 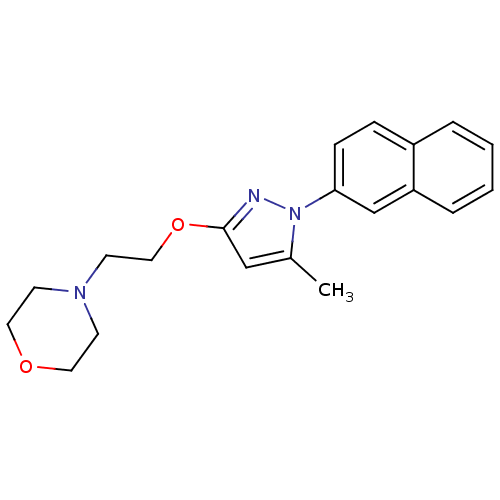
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Díaz, JL; Cuberes, R; Berrocal, J; Contijoch, M; Christmann, U; Fernández, A; Port, A; Holenz, J; Buschmann, H; Laggner, C; Serafini, MT; Burgueño, J; Zamanillo, D; Merlos, M; Vela, JM; Almansa, C Synthesis and biological evaluation of the 1-arylpyrazole class ofs(1) receptor antagonists: identification of 4-{2-[5-methyl-1-(naphthalen-2-yl)-1H-pyrazol-3-yloxy]ethyl}morpholine (S1RA, E-52862). J Med Chem55:8211-24 (2012) [PubMed] Article
Díaz, JL; Cuberes, R; Berrocal, J; Contijoch, M; Christmann, U; Fernández, A; Port, A; Holenz, J; Buschmann, H; Laggner, C; Serafini, MT; Burgueño, J; Zamanillo, D; Merlos, M; Vela, JM; Almansa, C Synthesis and biological evaluation of the 1-arylpyrazole class ofs(1) receptor antagonists: identification of 4-{2-[5-methyl-1-(naphthalen-2-yl)-1H-pyrazol-3-yloxy]ethyl}morpholine (S1RA, E-52862). J Med Chem55:8211-24 (2012) [PubMed] Article