| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cannabinoid receptor 1 |
|---|
| Ligand | BDBM50399111 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_876705 (CHEMBL2182818) |
|---|
| Ki | >20000±n/a nM |
|---|
| Citation |  Yang, P; Myint, KZ; Tong, Q; Feng, R; Cao, H; Almehizia, AA; Alqarni, MH; Wang, L; Bartlow, P; Gao, Y; Gertsch, J; Teramachi, J; Kurihara, N; Roodman, GD; Cheng, T; Xie, XQ Lead discovery, chemistry optimization, and biological evaluation studies of novel biamide derivatives as CB2 receptor inverse agonists and osteoclast inhibitors. J Med Chem55:9973-87 (2012) [PubMed] Article Yang, P; Myint, KZ; Tong, Q; Feng, R; Cao, H; Almehizia, AA; Alqarni, MH; Wang, L; Bartlow, P; Gao, Y; Gertsch, J; Teramachi, J; Kurihara, N; Roodman, GD; Cheng, T; Xie, XQ Lead discovery, chemistry optimization, and biological evaluation studies of novel biamide derivatives as CB2 receptor inverse agonists and osteoclast inhibitors. J Med Chem55:9973-87 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cannabinoid receptor 1 |
|---|
| Name: | Cannabinoid receptor 1 |
|---|
| Synonyms: | CANN6 | CANNABINOID CB1 | CB-R | CB1 | CNR | CNR1 | CNR1_HUMAN | Cannabinoid CB1 receptor | Cannabinoid receptor | Cannabinoid receptor 1 (CB1) | Cannabinoid receptor 1 (brain) |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52868.96 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P21554 |
|---|
| Residue: | 472 |
|---|
| Sequence: | MKSILDGLADTTFRTITTDLLYVGSNDIQYEDIKGDMASKLGYFPQKFPLTSFRGSPFQE
KMTAGDNPQLVPADQVNITEFYNKSLSSFKENEENIQCGENFMDIECFMVLNPSQQLAIA
VLSLTLGTFTVLENLLVLCVILHSRSLRCRPSYHFIGSLAVADLLGSVIFVYSFIDFHVF
HRKDSRNVFLFKLGGVTASFTASVGSLFLTAIDRYISIHRPLAYKRIVTRPKAVVAFCLM
WTIAIVIAVLPLLGWNCEKLQSVCSDIFPHIDETYLMFWIGVTSVLLLFIVYAYMYILWK
AHSHAVRMIQRGTQKSIIIHTSEDGKVQVTRPDQARMDIRLAKTLVLILVVLIICWGPLL
AIMVYDVFGKMNKLIKTVFAFCSMLCLLNSTVNPIIYALRSKDLRHAFRSMFPSCEGTAQ
PLDNSMGDSDCLHKHANNAASVHRAAESCIKSTVKIAKVTMSVSTDTSAEAL
|
|
|
|---|
| BDBM50399111 |
|---|
| n/a |
|---|
| Name | BDBM50399111 |
|---|
| Synonyms: | CHEMBL2179736 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C31H55N3O2 |
|---|
| Mol. Mass. | 501.7873 |
|---|
| SMILES | CCCCCCCCCC(=O)NC(NC(=O)CCCCCCCCC)c1ccc(cc1)N(CC)CC |
|---|
| Structure | 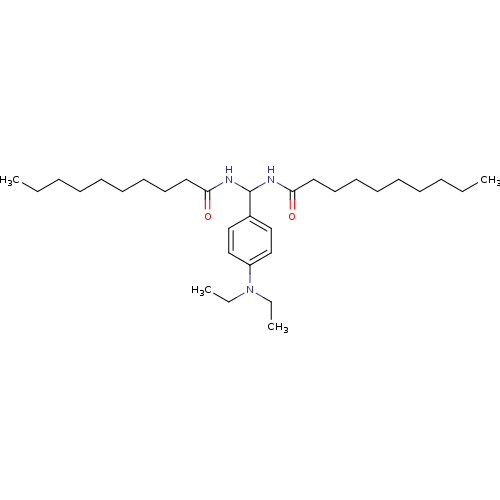
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Yang, P; Myint, KZ; Tong, Q; Feng, R; Cao, H; Almehizia, AA; Alqarni, MH; Wang, L; Bartlow, P; Gao, Y; Gertsch, J; Teramachi, J; Kurihara, N; Roodman, GD; Cheng, T; Xie, XQ Lead discovery, chemistry optimization, and biological evaluation studies of novel biamide derivatives as CB2 receptor inverse agonists and osteoclast inhibitors. J Med Chem55:9973-87 (2012) [PubMed] Article
Yang, P; Myint, KZ; Tong, Q; Feng, R; Cao, H; Almehizia, AA; Alqarni, MH; Wang, L; Bartlow, P; Gao, Y; Gertsch, J; Teramachi, J; Kurihara, N; Roodman, GD; Cheng, T; Xie, XQ Lead discovery, chemistry optimization, and biological evaluation studies of novel biamide derivatives as CB2 receptor inverse agonists and osteoclast inhibitors. J Med Chem55:9973-87 (2012) [PubMed] Article