| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 3A |
|---|
| Ligand | BDBM50399289 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_877121 (CHEMBL2187244) |
|---|
| IC50 | >31622.78±n/a nM |
|---|
| Citation |  Zanaletti, R; Bettinetti, L; Castaldo, C; Ceccarelli, I; Cocconcelli, G; Comery, TA; Dunlop, J; Genesio, E; Ghiron, C; Haydar, SN; Jow, F; Maccari, L; Micco, I; Nencini, A; Pratelli, C; Scali, C; Turlizzi, E; Valacchi, M N-[5-(5-fluoropyridin-3-yl)-1H-pyrazol-3-yl]-4-piperidin-1-ylbutyramide (SEN78702, WYE-308775): a medicinal chemistry effort toward ana7 nicotinic acetylcholine receptor agonist preclinical candidate. J Med Chem55:10277-81 (2012) [PubMed] Article Zanaletti, R; Bettinetti, L; Castaldo, C; Ceccarelli, I; Cocconcelli, G; Comery, TA; Dunlop, J; Genesio, E; Ghiron, C; Haydar, SN; Jow, F; Maccari, L; Micco, I; Nencini, A; Pratelli, C; Scali, C; Turlizzi, E; Valacchi, M N-[5-(5-fluoropyridin-3-yl)-1H-pyrazol-3-yl]-4-piperidin-1-ylbutyramide (SEN78702, WYE-308775): a medicinal chemistry effort toward ana7 nicotinic acetylcholine receptor agonist preclinical candidate. J Med Chem55:10277-81 (2012) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 3A |
|---|
| Name: | 5-hydroxytryptamine receptor 3A |
|---|
| Synonyms: | 5-HT-3 | 5-HT3 | 5-HT3A | 5-HT3R | 5-hydroxytryptamine receptor 3 (5-HT3) | 5-hydroxytryptamine receptor 3A (5-HT3a) | 5-hydroxytryptamine receptor 3A (5HT3A) | 5HT3A_HUMAN | 5HT3R | HTR3 | HTR3A | Serotonin 3 (5-HT3) receptor | Serotonin 3 receptor (5HT3) | Serotonin receptor 3A | Serotonin-gated ion channel receptor | Serotonin-gated ion channel receptor 3 |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 55283.27 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P46098 |
|---|
| Residue: | 478 |
|---|
| Sequence: | MLLWVQQALLALLLPTLLAQGEARRSRNTTRPALLRLSDYLLTNYRKGVRPVRDWRKPTT
VSIDVIVYAILNVDEKNQVLTTYIWYRQYWTDEFLQWNPEDFDNITKLSIPTDSIWVPDI
LINEFVDVGKSPNIPYVYIRHQGEVQNYKPLQVVTACSLDIYNFPFDVQNCSLTFTSWLH
TIQDINISLWRLPEKVKSDRSVFMNQGEWELLGVLPYFREFSMESSNYYAEMKFYVVIRR
RPLFYVVSLLLPSIFLMVMDIVGFYLPPNSGERVSFKITLLLGYSVFLIIVSDTLPATAI
GTPLIGVYFVVCMALLVISLAETIFIVRLVHKQDLQQPVPAWLRHLVLERIAWLLCLREQ
STSQRPPATSQATKTDDCSAMGNHCSHMGGPQDFEKSPRDRCSPPPPPREASLAVCGLLQ
ELSSIRQFLEKRDEIREVARDWLRVGSVLDKLLFHIYLLAVLAYSITLVMLWSIWQYA
|
|
|
|---|
| BDBM50399289 |
|---|
| n/a |
|---|
| Name | BDBM50399289 |
|---|
| Synonyms: | CHEMBL2180835 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H26N4O2 |
|---|
| Mol. Mass. | 342.4353 |
|---|
| SMILES | COc1ccc(cc1)-c1cc(NC(=O)CCCN2CCCCC2)[nH]n1 |
|---|
| Structure | 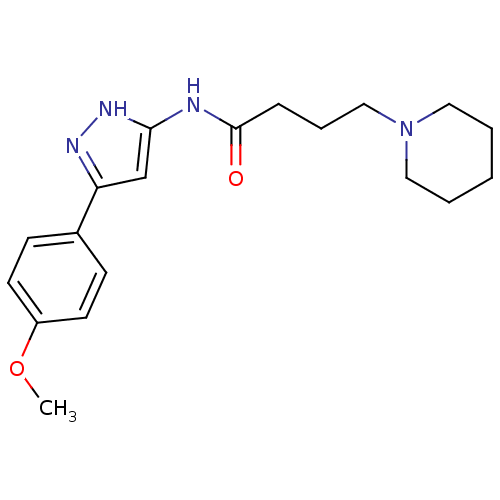
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Zanaletti, R; Bettinetti, L; Castaldo, C; Ceccarelli, I; Cocconcelli, G; Comery, TA; Dunlop, J; Genesio, E; Ghiron, C; Haydar, SN; Jow, F; Maccari, L; Micco, I; Nencini, A; Pratelli, C; Scali, C; Turlizzi, E; Valacchi, M N-[5-(5-fluoropyridin-3-yl)-1H-pyrazol-3-yl]-4-piperidin-1-ylbutyramide (SEN78702, WYE-308775): a medicinal chemistry effort toward ana7 nicotinic acetylcholine receptor agonist preclinical candidate. J Med Chem55:10277-81 (2012) [PubMed] Article
Zanaletti, R; Bettinetti, L; Castaldo, C; Ceccarelli, I; Cocconcelli, G; Comery, TA; Dunlop, J; Genesio, E; Ghiron, C; Haydar, SN; Jow, F; Maccari, L; Micco, I; Nencini, A; Pratelli, C; Scali, C; Turlizzi, E; Valacchi, M N-[5-(5-fluoropyridin-3-yl)-1H-pyrazol-3-yl]-4-piperidin-1-ylbutyramide (SEN78702, WYE-308775): a medicinal chemistry effort toward ana7 nicotinic acetylcholine receptor agonist preclinical candidate. J Med Chem55:10277-81 (2012) [PubMed] Article