| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2A |
|---|
| Ligand | BDBM50407361 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_2708 (CHEMBL617955) |
|---|
| IC50 | >10000±n/a nM |
|---|
| Citation |  Street, LJ; Baker, R; Davey, WB; Guiblin, AR; Jelley, RA; Reeve, AJ; Routledge, H; Sternfeld, F; Watt, AP; Beer, MS Synthesis and serotonergic activity of N,N-dimethyl-2-[5-(1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethylamine and analogues: potent agonists for 5-HT1D receptors. J Med Chem38:1799-810 (1995) [PubMed] Street, LJ; Baker, R; Davey, WB; Guiblin, AR; Jelley, RA; Reeve, AJ; Routledge, H; Sternfeld, F; Watt, AP; Beer, MS Synthesis and serotonergic activity of N,N-dimethyl-2-[5-(1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethylamine and analogues: potent agonists for 5-HT1D receptors. J Med Chem38:1799-810 (1995) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2A |
|---|
| Name: | 5-hydroxytryptamine receptor 2A |
|---|
| Synonyms: | 5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 52852.05 |
|---|
| Organism: | Rattus norvegicus (rat) |
|---|
| Description: | Rat cortex membranes 5-HT2A receptors. |
|---|
| Residue: | 471 |
|---|
| Sequence: | MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGY
LPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIAD
MLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNP
IHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSF
VAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIH
REPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGA
LLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYK
SSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
|
|
|
|---|
| BDBM50407361 |
|---|
| n/a |
|---|
| Name | BDBM50407361 |
|---|
| Synonyms: | CHEMBL53764 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C16H20N4 |
|---|
| Mol. Mass. | 268.3568 |
|---|
| SMILES | CN(C)CCc1c[nH]c2ccc(cc12)-n1ccnc1C |
|---|
| Structure | 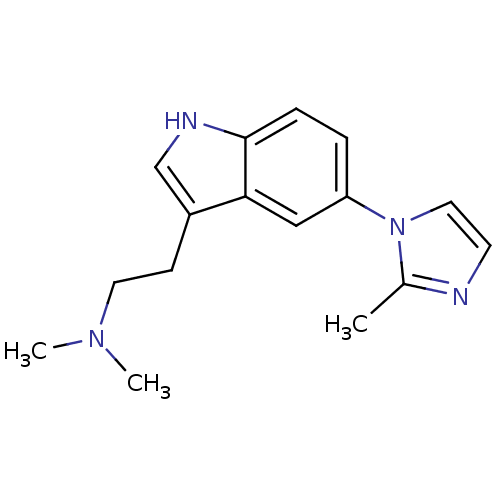
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Street, LJ; Baker, R; Davey, WB; Guiblin, AR; Jelley, RA; Reeve, AJ; Routledge, H; Sternfeld, F; Watt, AP; Beer, MS Synthesis and serotonergic activity of N,N-dimethyl-2-[5-(1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethylamine and analogues: potent agonists for 5-HT1D receptors. J Med Chem38:1799-810 (1995) [PubMed]
Street, LJ; Baker, R; Davey, WB; Guiblin, AR; Jelley, RA; Reeve, AJ; Routledge, H; Sternfeld, F; Watt, AP; Beer, MS Synthesis and serotonergic activity of N,N-dimethyl-2-[5-(1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethylamine and analogues: potent agonists for 5-HT1D receptors. J Med Chem38:1799-810 (1995) [PubMed]