| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 1A |
|---|
| Ligand | BDBM69602 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_539783 (CHEMBL1035001) |
|---|
| Ki | 2.09±n/a nM |
|---|
| Citation |  Quaglia, W; Piergentili, A; Del Bello, F; Farande, Y; Giannella, M; Pigini, M; Rafaiani, G; Carrieri, A; Amantini, C; Lucciarini, R; Santoni, G; Poggesi, E; Leonardi, A Structure-activity relationships in 1,4-benzodioxan-related compounds. 9. From 1,4-benzodioxane to 1,4-dioxane ring as a promising template of novel alpha1D-adrenoreceptor antagonists, 5-HT1A full agonists, and cytotoxic agents. J Med Chem51:6359-70 (2008) [PubMed] Article Quaglia, W; Piergentili, A; Del Bello, F; Farande, Y; Giannella, M; Pigini, M; Rafaiani, G; Carrieri, A; Amantini, C; Lucciarini, R; Santoni, G; Poggesi, E; Leonardi, A Structure-activity relationships in 1,4-benzodioxan-related compounds. 9. From 1,4-benzodioxane to 1,4-dioxane ring as a promising template of novel alpha1D-adrenoreceptor antagonists, 5-HT1A full agonists, and cytotoxic agents. J Med Chem51:6359-70 (2008) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 1A |
|---|
| Name: | 5-hydroxytryptamine receptor 1A |
|---|
| Synonyms: | 5-HT-1A | 5-HT1A | 5-hydroxytryptamine receptor 1A (5-HT-1A) | 5HT1A_HUMAN | ADRB2RL1 | ADRBRL1 | Dopamine D2 receptor and serotonin 1a receptor | G-21 | HTR1A | Serotonin receptor 1A |
|---|
| Type: | n/a |
|---|
| Mol. Mass.: | 46122.49 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | n/a |
|---|
| Residue: | 422 |
|---|
| Sequence: | MDVLSPGQGNNTTSPPAPFETGGNTTGISDVTVSYQVITSLLLGTLIFCAVLGNACVVAA
IALERSLQNVANYLIGSLAVTDLMVSVLVLPMAALYQVLNKWTLGQVTCDLFIALDVLCC
TSSILHLCAIALDRYWAITDPIDYVNKRTPRRAAALISLTWLIGFLISIPPMLGWRTPED
RSDPDACTISKDHGYTIYSTFGAFYIPLLLMLVLYGRIFRAARFRIRKTVKKVEKTGADT
RHGASPAPQPKKSVNGESGSRNWRLGVESKAGGALCANGAVRQGDDGAALEVIEVHRVGN
SKEHLPLPSEAGPTPCAPASFERKNERNAEAKRKMALARERKTVKTLGIIMGTFILCWLP
FFIVALVLPFCESSCHMPTLLGAIINWLGYSNSLLNPVIYAYFNKDFQNAFKKIIKCKFC
RQ
|
|
|
|---|
| BDBM69602 |
|---|
| n/a |
|---|
| Name | BDBM69602 |
|---|
| Synonyms: | 2,3-dihydro-1,4-benzodioxin-3-ylmethyl-[2-(2,6-dimethoxyphenoxy)ethyl]amine;hydrochloride | CHEMBL25554 | MLS000859914 | N-(2,3-dihydro-1,4-benzodioxin-3-ylmethyl)-2-(2,6-dimethoxyphenoxy)ethanamine;hydrochloride | SMR000326775 | WB-4101 | WB-4101 hydrochloride | cid_11957505 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H23NO5 |
|---|
| Mol. Mass. | 345.3896 |
|---|
| SMILES | COc1cccc(OC)c1OCCNCC1COc2ccccc2O1 |
|---|
| Structure | 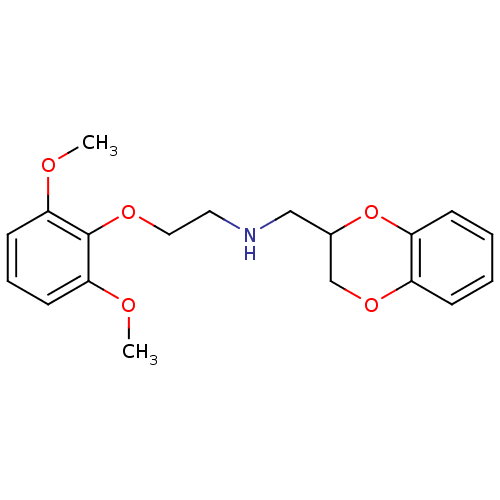
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Quaglia, W; Piergentili, A; Del Bello, F; Farande, Y; Giannella, M; Pigini, M; Rafaiani, G; Carrieri, A; Amantini, C; Lucciarini, R; Santoni, G; Poggesi, E; Leonardi, A Structure-activity relationships in 1,4-benzodioxan-related compounds. 9. From 1,4-benzodioxane to 1,4-dioxane ring as a promising template of novel alpha1D-adrenoreceptor antagonists, 5-HT1A full agonists, and cytotoxic agents. J Med Chem51:6359-70 (2008) [PubMed] Article
Quaglia, W; Piergentili, A; Del Bello, F; Farande, Y; Giannella, M; Pigini, M; Rafaiani, G; Carrieri, A; Amantini, C; Lucciarini, R; Santoni, G; Poggesi, E; Leonardi, A Structure-activity relationships in 1,4-benzodioxan-related compounds. 9. From 1,4-benzodioxane to 1,4-dioxane ring as a promising template of novel alpha1D-adrenoreceptor antagonists, 5-HT1A full agonists, and cytotoxic agents. J Med Chem51:6359-70 (2008) [PubMed] Article