| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A5 |
|---|
| Ligand | BDBM520 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_738406 (CHEMBL1743483) |
|---|
| Ki | 120±n/a nM |
|---|
| Citation |  Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed] Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A5 |
|---|
| Name: | Cytochrome P450 3A5 |
|---|
| Synonyms: | CP3A5_HUMAN | CYP3A5 | Cytochrome P450 3A5 | Cytochrome P450 3A5 (CYP3A5) |
|---|
| Type: | Protein |
|---|
| Mol. Mass.: | 57118.00 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P20815 |
|---|
| Residue: | 502 |
|---|
| Sequence: | MDLIPNLAVETWLLLAVSLVLLYLYGTRTHGLFKRLGIPGPTPLPLLGNVLSYRQGLWKF
DTECYKKYGKMWGTYEGQLPVLAITDPDVIRTVLVKECYSVFTNRRSLGPVGFMKSAISL
AEDEEWKRIRSLLSPTFTSGKLKEMFPIIAQYGDVLVRNLRREAEKGKPVTLKDIFGAYS
MDVITGTSFGVNIDSLNNPQDPFVESTKKFLKFGFLDPLFLSIILFPFLTPVFEALNVSL
FPKDTINFLSKSVNRMKKSRLNDKQKHRLDFLQLMIDSQNSKETESHKALSDLELAAQSI
IFIFAGYETTSSVLSFTLYELATHPDVQQKLQKEIDAVLPNKAPPTYDAVVQMEYLDMVV
NETLRLFPVAIRLERTCKKDVEINGVFIPKGSMVVIPTYALHHDPKYWTEPEEFRPERFS
KKKDSIDPYIYTPFGTGPRNCIGMRFALMNMKLALIRVLQNFSFKPCKETQIPLKLDTQG
LLQPEKPIVLKVDSRDGTLSGE
|
|
|
|---|
| BDBM520 |
|---|
| n/a |
|---|
| Name | BDBM520 |
|---|
| Synonyms: | 1,3-thiazol-5-ylmethyl N-[(2S,3S,5S)-3-hydroxy-5-[(2S)-3-methyl-2-{[methyl({[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl})carbamoyl]amino}butanamido]-1,6-diphenylhexan-2-yl]carbamate | ABT-538 | CHEMBL163 | Norvir | RTV | Ritonavir |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C37H48N6O5S2 |
|---|
| Mol. Mass. | 720.944 |
|---|
| SMILES | CC(C)[C@H](NC(=O)N(C)Cc1csc(n1)C(C)C)C(=O)N[C@H](C[C@H](O)[C@H](Cc1ccccc1)NC(=O)OCc1cncs1)Cc1ccccc1 |
|---|
| Structure | 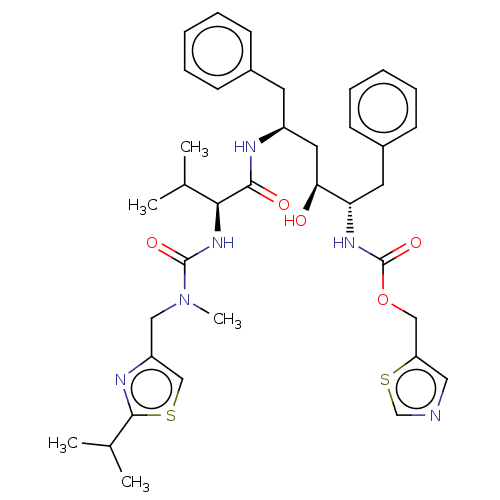
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed]
Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed]