| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Cytochrome P450 3A6 |
|---|
| Ligand | BDBM50014323 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_738411 (CHEMBL1743488) |
|---|
| Ki | 80100±n/a nM |
|---|
| Citation |  Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed] Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed] |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Cytochrome P450 3A6 |
|---|
| Name: | Cytochrome P450 3A6 |
|---|
| Synonyms: | CP3A6_RABIT | CYP3A6 | CYPIIIA6 | Cytochrome P450-3C |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 57463.93 |
|---|
| Organism: | Oryctolagus cuniculus |
|---|
| Description: | ChEMBL_738411 |
|---|
| Residue: | 501 |
|---|
| Sequence: | MDLIFSLETWVLLAASLVLLYLYGTSTHGLFKKMGIPGPTPLPFIGTILEYRKGIWDFDI
ECRKKYGKMWGLFDGRQPLMVITDPDMIKTVLVKECYSVFTNRRSFGPVGFMKKAVSISE
DEDWKRVRTLLSPTFTSGKLKEMLPIIAQYGDVLVKNLRQEAEKGKPVDLKEIFGAYSMD
VITGTSFGVNIDSLRNPQDPFVKNVRRLLKFSFFDPLLLSITLFPFLTPIFEALHISMFP
KDVMDFLKTSVEKIKDDRLKDKQKRRVDFLQLMINSQNSKEIDSHKALDDIEVVAQSIII
LFAGYETTSSTLSFIMHLLATHPDVQQKLQEEIDTLLPNKELATYDTLVKMEYLDMVVNE
TLRLYPIAGRLERVCKKDVDINGTFIPKGTIVMMPTYALHRDPQHWTEPDEFRPERFSKK
NKDNINPYIYHPFGAGPRNCLGMRFALMNIKLALVRLMQNFSFKLCKETQVPLKLGKQGL
LQPEKPIVLKVVSRDGIIRGA
|
|
|
|---|
| BDBM50014323 |
|---|
| n/a |
|---|
| Name | BDBM50014323 |
|---|
| Synonyms: | 2-PHENYL-4H-BENZO[H]CHROMEN-4-ONE | 2-Phenyl-benzo[h]chromen-4-one | 7,8-Benzoflavone | CHEMBL283196 | alpha-naphthoflavone |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C19H12O2 |
|---|
| Mol. Mass. | 272.2974 |
|---|
| SMILES | O=c1cc(oc2c3ccccc3ccc12)-c1ccccc1 |
|---|
| Structure | 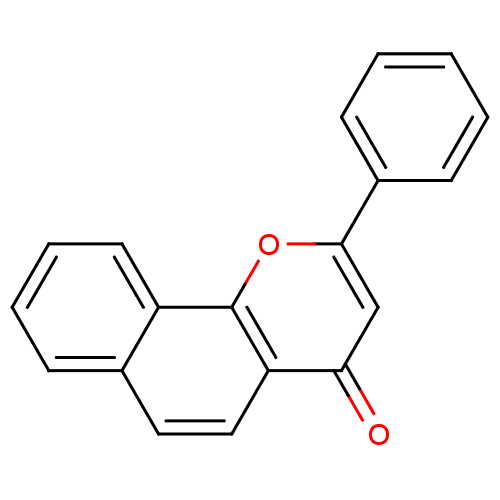
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed]
Fontana, E; Dansette, PM; Poli, SM Cytochrome p450 enzymes mechanism based inhibitors: common sub-structures and reactivity. Curr Drug Metab6:413-54 (2005) [PubMed]