| Reaction Details |
|---|
 | Report a problem with these data |
| Target | 5-hydroxytryptamine receptor 2C |
|---|
| Ligand | BDBM31005 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_946114 (CHEMBL2343042) |
|---|
| Ki | 1.7±n/a nM |
|---|
| Citation |  Jensen, AA; Plath, N; Pedersen, MH; Isberg, V; Krall, J; Wellendorph, P; Stensbøl, TB; Gloriam, DE; Krogsgaard-Larsen, P; Frølund, B Design, synthesis, and pharmacological characterization of N- and O-substituted 5,6,7,8-tetrahydro-4H-isoxazolo[4,5-d]azepin-3-ol analogues: novel 5-HT(2A)/5-HT(2C) receptor agonists with pro-cognitive properties. J Med Chem56:1211-27 (2013) [PubMed] Article Jensen, AA; Plath, N; Pedersen, MH; Isberg, V; Krall, J; Wellendorph, P; Stensbøl, TB; Gloriam, DE; Krogsgaard-Larsen, P; Frølund, B Design, synthesis, and pharmacological characterization of N- and O-substituted 5,6,7,8-tetrahydro-4H-isoxazolo[4,5-d]azepin-3-ol analogues: novel 5-HT(2A)/5-HT(2C) receptor agonists with pro-cognitive properties. J Med Chem56:1211-27 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| 5-hydroxytryptamine receptor 2C |
|---|
| Name: | 5-hydroxytryptamine receptor 2C |
|---|
| Synonyms: | 5-HT-1C | 5-HT-2C | 5-HT1C | 5-HT2C | 5-HT2C-INI | 5-HT2c VGI | 5-HTR2C | 5-hydroxytryptamine receptor 1C | 5-hydroxytryptamine receptor 2C (5-HT-2C) | 5-hydroxytryptamine receptor 2C (5HT-2C) | 5HT-1C | 5HT2C_HUMAN | HTR1C | HTR2C | Serotonin (5-HT3) receptor | Serotonin 2c (5-HT2c) receptor | Serotonin Receptor 2C |
|---|
| Type: | G Protein-Coupled Receptor (GPCR) |
|---|
| Mol. Mass.: | 51836.79 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P28335 |
|---|
| Residue: | 458 |
|---|
| Sequence: | MVNLRNAVHSFLVHLIGLLVWQSDISVSPVAAIVTDIFNTSDGGRFKFPDGVQNWPALSI
VIIIIMTIGGNILVIMAVSMEKKLHNATNYFLMSLAIADMLVGLLVMPLSLLAILYDYVW
PLPRYLCPVWISLDVLFSTASIMHLCAISLDRYVAIRNPIEHSRFNSRTKAIMKIAIVWA
ISIGVSVPIPVIGLRDEEKVFVNNTTCVLNDPNFVLIGSFVAFFIPLTIMVITYCLTIYV
LRRQALMLLHGHTEEPPGLSLDFLKCCKRNTAEEENSANPNQDQNARRRKKKERRPRGTM
QAINNERKASKVLGIVFFVFLIMWCPFFITNILSVLCEKSCNQKLMEKLLNVFVWIGYVC
SGINPLVYTLFNKIYRRAFSNYLRCNYKVEKKPPVRQIPRVAATALSGRELNVNIYRHTN
EPVIEKASDNEPGIEMQVENLELPVNPSSVVSERISSV
|
|
|
|---|
| BDBM31005 |
|---|
| n/a |
|---|
| Name | BDBM31005 |
|---|
| Synonyms: | 2-methyl-1,2,3,4,10,14b-hexahydrodibenzo[c,f]pyrazino[1,2-a]azepine | MIANSERIN HYDROCHLORIDE | MLS000069681 | SMR000058472 | cid_68551 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H21ClN2 |
|---|
| Mol. Mass. | 300.826 |
|---|
| SMILES | Cl.CN1CCN2C(C1)c1ccccc1Cc1ccccc21 |
|---|
| Structure | 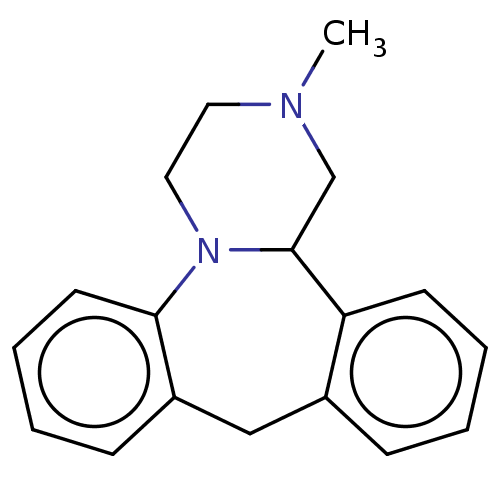
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Jensen, AA; Plath, N; Pedersen, MH; Isberg, V; Krall, J; Wellendorph, P; Stensbøl, TB; Gloriam, DE; Krogsgaard-Larsen, P; Frølund, B Design, synthesis, and pharmacological characterization of N- and O-substituted 5,6,7,8-tetrahydro-4H-isoxazolo[4,5-d]azepin-3-ol analogues: novel 5-HT(2A)/5-HT(2C) receptor agonists with pro-cognitive properties. J Med Chem56:1211-27 (2013) [PubMed] Article
Jensen, AA; Plath, N; Pedersen, MH; Isberg, V; Krall, J; Wellendorph, P; Stensbøl, TB; Gloriam, DE; Krogsgaard-Larsen, P; Frølund, B Design, synthesis, and pharmacological characterization of N- and O-substituted 5,6,7,8-tetrahydro-4H-isoxazolo[4,5-d]azepin-3-ol analogues: novel 5-HT(2A)/5-HT(2C) receptor agonists with pro-cognitive properties. J Med Chem56:1211-27 (2013) [PubMed] Article