| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Coagulation factor XI |
|---|
| Ligand | BDBM50431213 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_948831 (CHEMBL2340213) |
|---|
| Ki | >120000±n/a nM |
|---|
| Citation |  Baeriswyl, V; Calzavarini, S; Gerschheimer, C; Diderich, P; Angelillo-Scherrer, A; Heinis, C Development of a selective peptide macrocycle inhibitor of coagulation factor XII toward the generation of a safe antithrombotic therapy. J Med Chem56:3742-6 (2013) [PubMed] Article Baeriswyl, V; Calzavarini, S; Gerschheimer, C; Diderich, P; Angelillo-Scherrer, A; Heinis, C Development of a selective peptide macrocycle inhibitor of coagulation factor XII toward the generation of a safe antithrombotic therapy. J Med Chem56:3742-6 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Coagulation factor XI |
|---|
| Name: | Coagulation factor XI |
|---|
| Synonyms: | Coagulation factor XIa | Coagulation factor XIa heavy chain | Coagulation factor XIa light chain | F11 | FA11_HUMAN | FXI | Factor XIa | Factor XIa (fXIa) | PTA | Plasma thromboplastin antecedent |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 70130.58 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P03951 |
|---|
| Residue: | 625 |
|---|
| Sequence: | MIFLYQVVHFILFTSVSGECVTQLLKDTCFEGGDITTVFTPSAKYCQVVCTYHPRCLLFT
FTAESPSEDPTRWFTCVLKDSVTETLPRVNRTAAISGYSFKQCSHQISACNKDIYVDLDM
KGINYNSSVAKSAQECQERCTDDVHCHFFTYATRQFPSLEHRNICLLKHTQTGTPTRITK
LDKVVSGFSLKSCALSNLACIRDIFPNTVFADSNIDSVMAPDAFVCGRICTHHPGCLFFT
FFSQEWPKESQRNLCLLKTSESGLPSTRIKKSKALSGFSLQSCRHSIPVFCHSSFYHDTD
FLGEELDIVAAKSHEACQKLCTNAVRCQFFTYTPAQASCNEGKGKCYLKLSSNGSPTKIL
HGRGGISGYTLRLCKMDNECTTKIKPRIVGGTASVRGEWPWQVTLHTTSPTQRHLCGGSI
IGNQWILTAAHCFYGVESPKILRVYSGILNQSEIKEDTSFFGVQEIIIHDQYKMAESGYD
IALLKLETTVNYTDSQRPICLPSKGDRNVIYTDCWVTGWGYRKLRDKIQNTLQKAKIPLV
TNEECQKRYRGHKITHKMICAGYREGGKDACKGDSGGPLSCKHNEVWHLVGITSWGEGCA
QRERPGVYTNVVEYVDWILEKTQAV
|
|
|
|---|
| BDBM50431213 |
|---|
| n/a |
|---|
| Name | BDBM50431213 |
|---|
| Synonyms: | CHEMBL2332604 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C59H82N16O15S3 |
|---|
| Mol. Mass. | 1351.576 |
|---|
| SMILES | [#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@@H]-2-[#6]-[#6]-[#6]-[#7]-2-[#6](=O)-[#6@@H]-2-[#6]-[#16]-[#6]-c3cc(-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-c4ccc(-[#8])cc4)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-[#6]-[#6](-[#8])=O)cc(-[#6]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6]-[#7])-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6]-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-2)c3 |r| |
|---|
| Structure | 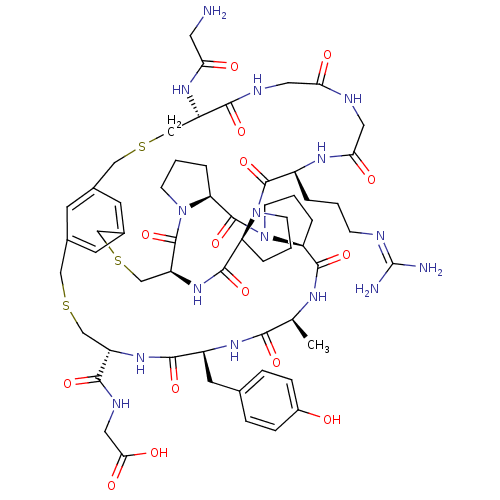
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Baeriswyl, V; Calzavarini, S; Gerschheimer, C; Diderich, P; Angelillo-Scherrer, A; Heinis, C Development of a selective peptide macrocycle inhibitor of coagulation factor XII toward the generation of a safe antithrombotic therapy. J Med Chem56:3742-6 (2013) [PubMed] Article
Baeriswyl, V; Calzavarini, S; Gerschheimer, C; Diderich, P; Angelillo-Scherrer, A; Heinis, C Development of a selective peptide macrocycle inhibitor of coagulation factor XII toward the generation of a safe antithrombotic therapy. J Med Chem56:3742-6 (2013) [PubMed] Article