| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Methionine aminopeptidase 2 |
|---|
| Ligand | BDBM50433679 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_959306 (CHEMBL2383873) |
|---|
| IC50 | 2080±n/a nM |
|---|
| Citation |  Sun, J; Li, MH; Qian, SS; Guo, FJ; Dang, XF; Wang, XM; Xue, YR; Zhu, HL Synthesis and antitumor activity of 1,3,4-oxadiazole possessing 1,4-benzodioxan moiety as a novel class of potent methionine aminopeptidase type II inhibitors. Bioorg Med Chem Lett23:2876-9 (2013) [PubMed] Article Sun, J; Li, MH; Qian, SS; Guo, FJ; Dang, XF; Wang, XM; Xue, YR; Zhu, HL Synthesis and antitumor activity of 1,3,4-oxadiazole possessing 1,4-benzodioxan moiety as a novel class of potent methionine aminopeptidase type II inhibitors. Bioorg Med Chem Lett23:2876-9 (2013) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Methionine aminopeptidase 2 |
|---|
| Name: | Methionine aminopeptidase 2 |
|---|
| Synonyms: | Initiation factor 2-associated 67 kDa glycoprotein | MAP 2 | MAP2_HUMAN | METAP2 | MNPEP | MetAP 2 | Methionine aminopeptidase 2 (MetAP2) | Methionine aminopeptidases (HsMetAP2) | P67EIF2 | Peptidase M 2 | p67 |
|---|
| Type: | Enzyme |
|---|
| Mol. Mass.: | 52884.45 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | P50579 |
|---|
| Residue: | 478 |
|---|
| Sequence: | MAGVEEVAASGSHLNGDLDPDDREEGAASTAEEAAKKKRRKKKKSKGPSAAGEQEPDKES
GASVDEVARQLERSALEDKERDEDDEDGDGDGDGATGKKKKKKKKKRGPKVQTDPPSVPI
CDLYPNGVFPKGQECEYPPTQDGRTAAWRTTSEEKKALDQASEEIWNDFREAAEAHRQVR
KYVMSWIKPGMTMIEICEKLEDCSRKLIKENGLNAGLAFPTGCSLNNCAAHYTPNAGDTT
VLQYDDICKIDFGTHISGRIIDCAFTVTFNPKYDTLLKAVKDATNTGIKCAGIDVRLCDV
GEAIQEVMESYEVEIDGKTYQVKPIRNLNGHSIGQYRIHAGKTVPIVKGGEATRMEEGEV
YAIETFGSTGKGVVHDDMECSHYMKNFDVGHVPIRLPRTKHLLNVINENFGTLAFCRRWL
DRLGESKYLMALKNLCDLGIVDPYPPLCDIKGSYTAQFEHTILLRPTCKEVVSRGDDY
|
|
|
|---|
| BDBM50433679 |
|---|
| n/a |
|---|
| Name | BDBM50433679 |
|---|
| Synonyms: | CHEMBL2381120 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C18H13FN2O3 |
|---|
| Mol. Mass. | 324.3058 |
|---|
| SMILES | Fc1ccccc1\C=C\c1nnc(o1)-c1ccc2OCCOc2c1 |
|---|
| Structure | 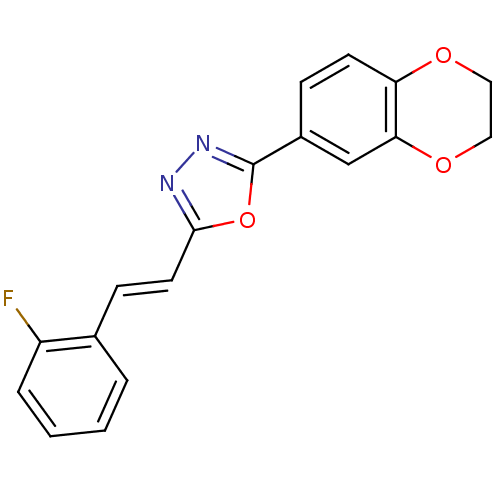
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Sun, J; Li, MH; Qian, SS; Guo, FJ; Dang, XF; Wang, XM; Xue, YR; Zhu, HL Synthesis and antitumor activity of 1,3,4-oxadiazole possessing 1,4-benzodioxan moiety as a novel class of potent methionine aminopeptidase type II inhibitors. Bioorg Med Chem Lett23:2876-9 (2013) [PubMed] Article
Sun, J; Li, MH; Qian, SS; Guo, FJ; Dang, XF; Wang, XM; Xue, YR; Zhu, HL Synthesis and antitumor activity of 1,3,4-oxadiazole possessing 1,4-benzodioxan moiety as a novel class of potent methionine aminopeptidase type II inhibitors. Bioorg Med Chem Lett23:2876-9 (2013) [PubMed] Article