| Reaction Details |
|---|
 | Report a problem with these data |
| Target | Neuroendocrine convertase 2 |
|---|
| Ligand | BDBM50446944 |
|---|
| Substrate/Competitor | n/a |
|---|
| Meas. Tech. | ChEMBL_1290738 (CHEMBL3117750) |
|---|
| Ki | 0.240000±n/a nM |
|---|
| Citation |  Kwiatkowska, A; Couture, F; Levesque, C; Ly, K; Desjardins, R; Beauchemin, S; Prahl, A; Lammek, B; Neugebauer, W; Dory, YL; Day, R Design, synthesis, and structure-activity relationship studies of a potent PACE4 inhibitor. J Med Chem57:98-109 (2014) [PubMed] Article Kwiatkowska, A; Couture, F; Levesque, C; Ly, K; Desjardins, R; Beauchemin, S; Prahl, A; Lammek, B; Neugebauer, W; Dory, YL; Day, R Design, synthesis, and structure-activity relationship studies of a potent PACE4 inhibitor. J Med Chem57:98-109 (2014) [PubMed] Article |
|---|
| More Info.: | Get all data from this article, Assay Method |
|---|
| |
| Neuroendocrine convertase 2 |
|---|
| Name: | Neuroendocrine convertase 2 |
|---|
| Synonyms: | KEX2-like endoprotease 2 | NEC 2 | NEC2 | NEC2_HUMAN | Neuroendocrine convertase 2 | PC2 | PCSK2 | Prohormone convertase 2 | Proprotein convertase 2 |
|---|
| Type: | PROTEIN |
|---|
| Mol. Mass.: | 70563.93 |
|---|
| Organism: | Homo sapiens (Human) |
|---|
| Description: | ChEMBL_1290738 |
|---|
| Residue: | 638 |
|---|
| Sequence: | MKGGCVSQWKAAAGFLFCVMVFASAERPVFTNHFLVELHKGGEDKARQVAAEHGFGVRKL
PFAEGLYHFYHNGLAKAKRRRSLHHKQQLERDPRVKMALQQEGFDRKKRGYRDINEIDIN
MNDPLFTKQWYLINTGQADGTPGLDLNVAEAWELGYTGKGVTIGIMDDGIDYLHPDLASN
YNAEASYDFSSNDPYPYPRYTDDWFNSHGTRCAGEVSAAANNNICGVGVAYNSKVAGIRM
LDQPFMTDIIEASSISHMPQLIDIYSASWGPTDNGKTVDGPRELTLQAMADGVNKGRGGK
GSIYVWASGDGGSYDDCNCDGYASSMWTISINSAINDGRTALYDESCSSTLASTFSNGRK
RNPEAGVATTDLYGNCTLRHSGTSAAAPEAAGVFALALEANLGLTWRDMQHLTVLTSKRN
QLHDEVHQWRRNGVGLEFNHLFGYGVLDAGAMVKMAKDWKTVPERFHCVGGSVQDPEKIP
STGKLVLTLTTDACEGKENFVRYLEHVQAVITVNATRRGDLNINMTSPMGTKSILLSRRP
RDDDSKVGFDKWPFMTTHTWGEDARGTWTLELGFVGSAPQKGVLKEWTLMLHGTQSAPYI
DQVVRDYQSKLAMSKKEELEEELDEAVERSLKSILNKN
|
|
|
|---|
| BDBM50446944 |
|---|
| n/a |
|---|
| Name | BDBM50446944 |
|---|
| Synonyms: | CHEMBL3115771 |
|---|
| Type | Small organic molecule |
|---|
| Emp. Form. | C51H90N14O8 |
|---|
| Mol. Mass. | 1027.3493 |
|---|
| SMILES | [#6]-[#6](-[#6])-[#6]-[#6@H](-[#7]-[#6](-[#6])=O)-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6]-c1ccc(cc1)-[#6](-[#7])=[#7] |r| |
|---|
| Structure | 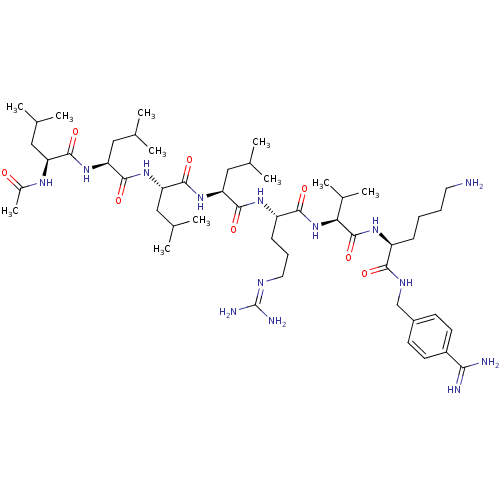
|
|---|


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data

 Kwiatkowska, A; Couture, F; Levesque, C; Ly, K; Desjardins, R; Beauchemin, S; Prahl, A; Lammek, B; Neugebauer, W; Dory, YL; Day, R Design, synthesis, and structure-activity relationship studies of a potent PACE4 inhibitor. J Med Chem57:98-109 (2014) [PubMed] Article
Kwiatkowska, A; Couture, F; Levesque, C; Ly, K; Desjardins, R; Beauchemin, S; Prahl, A; Lammek, B; Neugebauer, W; Dory, YL; Day, R Design, synthesis, and structure-activity relationship studies of a potent PACE4 inhibitor. J Med Chem57:98-109 (2014) [PubMed] Article